Quantitative Reverse transcription PCR (RT-qPCR) is frequently used in the lab to detect and quantify RNA expression in a sample. The first step of the assay is to convert the labile RNA to its complementary DNA (cDNA) counterpart through reverse transcription (RT).
In fact, RT is the first step in a variety of molecular biology techniques used to study RNA, because converting the labile RNA to more stable DNA makes the analysis easier and more reliable.
One-Step vs Two-Step qPCR
When performing RT-qPCR, you can set up the reverse transcription reaction using two methods: the one-step and the two-step method.
Put this article into practice
Choose a free resource to help you move forward
DOWNLOAD
Blood Collection Tube Chart
POSTER
Antibiotics Reference Guide
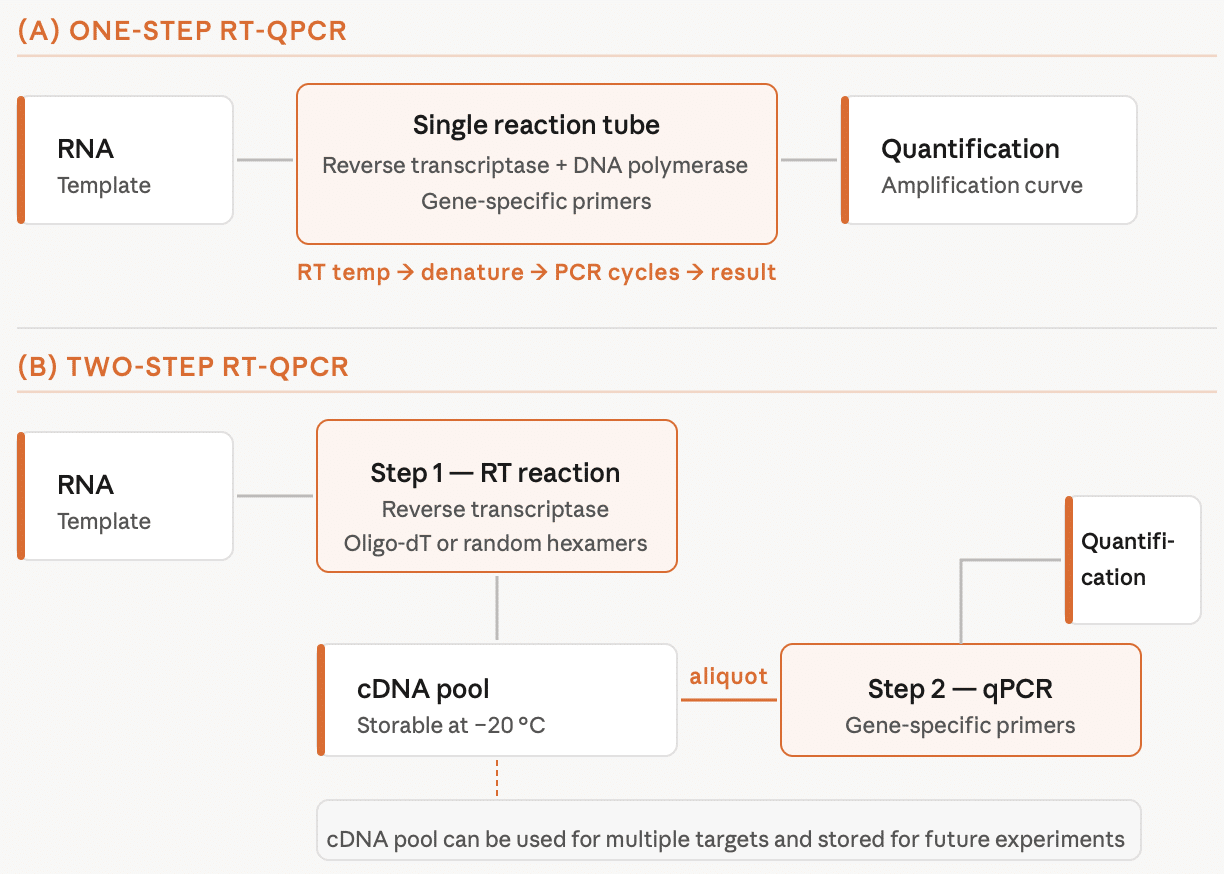
In one-step RT-qPCR (Figure 1a) both the RT reaction and qPCR amplification are performed in the same tube. The RNA template is added to the tube with two enzymes (reverse transcriptase and DNA polymerase) and all necessary components to complete the reaction. The reverse transcriptase generates the cDNA product, then the reverse transcriptase and cDNA are denatured and the DNA polymerase amplifies the cDNA. In the one-step method, the RT reaction is primed using a gene–specific primer. Therefore, only the region of interest is reverse-transcribed and subsequently amplified.
In two-step RT-qPCR, the RT reaction is performed separately from qPCR amplification (Figure 1b). First, RNA is added to the reaction mixture containing reverse transcriptase and oligo-dTs (that bind to the poly-A tails of mRNA) or random hexamers (or a combination of both) to synthesize cDNA. In the next step, an aliquot of the cDNA is removed from the tube, placed into a new tube and used as a template for qPCR with gene-specific primers. Because the RT reaction employs random priming or priming with oligo-dTs, either total RNA or total mRNA is transcribed in the RT step.
Although both methods should give you the same final result, there are advantages and disadvantages to each method. The decision on which method to choose depends on various factors. These factors are summarized in Table 1 and explained below.
Table.1 The Advantages and Disadvantages of One-step and Two-step RT-qPCR
One-step RT-PCR | Two-step RT-PCR | |
Pros | Fast | Easier to control |
Reduced chance of pipetting errors | Enables analysis of multiple targets | |
Less expensive for single targets | cDNA can be stored | |
Sensitive | ||
Cons | Less control over the process | Time-consuming |
Limited number of targets can be analyzed | Higher probability of contamination |
Considerations for One-Step RT-qPCR
Advantages: One-step RT-qPCR is faster to perform than two-step RT-qPCR, because the reaction occurs in a single tube. It also requires less preparation and hands-on time, which reduces the chances of pipetting errors, contamination, and similar sources of error.
Considerations: A disadvantage of this approach is that only a limited number of genes can be amplified from the same sample. Detecting several targets is possible with multiplexing (amplification of multiple targets simultaneously). However, multiplexing requires optimization using different primer pairs to decrease the chances of mispriming and primer-dimer formation. Another associated challenge lies in the compatibility of the components since the one-step method is a trade-off between transcription and amplification. For this reason, you will most likely need to use a kit for one-step RT-qPCR. This method also gives you less control over the process, which somewhat complicates troubleshooting.
Recommended for: Researchers usually decide to use one-step RT-qPCR when time is of the essence. For example, the speedy one-step RT-qPCR is a good approach for viral RNA detection when results must be acquired swiftly. It is also convenient for high throughput analysis and if you are using liquid handling robotics in your lab. Since this method employs gene specific primers, one-step RT-qPCR is often a better option for analyzing genes with low expression levels. Also, one-step qPCR, because it is very precise, is typically the method of choice when small differences in expression levels need to be measured.
Considerations for Two-Step RT-qPCR
Advantages: Two-step RT-qPCR enables analysis of multiple targets from the same sample. Because the reverse transcription step converts all the RNA from the sample into cDNA, the latter can be stored and used for later experiments. You can use the stored cDNA to detect a multitude of genes. A big benefit of the two-step method is the ability to individually optimize both the RT and qPCR steps, giving you more control over the process. Additionally, because only a small amount of the transcribed material is used for qPCR, any inhibitor that may have been carried over from RNA isolation to RT reaction (such as ethanol, phenol or guanidinium salt) is diluted.
Considerations: The obvious disadvantages to two-step RT-qPCR are that it is more time-consuming and has a higher possibility of contamination. Additionally, the two-step approach may introduce bias, because the RT step transcribes all RNA species to the same efficiency. When opting for this approach, you should take into consideration that priming influences the results of qPCR. For example, priming with oligo-dTs may not give the same result as priming using random hexamer primers. Therefore, you should use the same conditions for each RT reaction. In addition when using SYBR chemistry for detection, whether using one-step or two-step RT-qPCR, primer specificity is a concern. You can improve specificity in two-step reactions by using enhanced master mix solutions that contain enzymes and buffers that improve the specificity of the technique.
Recommended for: The most common reason for choosing the two-step method is the analysis of multiple targets from the same sample. Secondly, when storage of RNA is an issue, it is best to perform two-step RT-qPCR, because cDNA is stable at -20°C. Finally, use two-step RT-qPCR when the starting material is limited because two-step protocols generally provide higher cDNA yields.
Considerations for Both Methods
Although it may not seem obvious, the choice of RT enzyme component may significantly impact the results of RT-qPCR experiment. Different RT enzymes are active at different temperatures, and their performance may vary depending on the complexity of the starting material and the target sequence.
Why Is the RT Enzyme Affected by Temperature?
RT enzymes with greater thermostability enable use at elevated temperatures. These can help resolve RNA secondary structure and facilitate read-through of high GC content sections of the template. This, in turn, leads to higher cDNA yields, greater full-length cDNA generation, and better cDNA representation of the RNA population. In one-step RT-PCR protocols using gene-specific primers, enzyme use at elevated temperatures also improves primer specificity. For all of these reasons, thermostable RT mutant enzymes are a popular choice for both one-step and two-step RT-qPCR protocols.
Preparation is paramount! Take a few hours to thoroughly investigate which method is best for your experiment. Consider the sample type, level of gene expression, number of genes and samples to be analyzed. Plus the time you have to execute the analysis.
For more help and advice on RT, check out our article on the common pitfalls of reverse transcription.
References
T. Nolan, R. Hands & S. Bustin (2006) Quantification of mRNA using real-time RT-PCR. Nature Protocols, 1 (3): 1559- 1582
M. Wacker & M. Godard (2005) Analysis of One-Step and Two-Step Real-Time RT-PCR Using SuperScript III. J Biomol Tech, 16 (3): 266–271
You made it to the end—nice work! If you’re the kind of scientist who likes figuring things out without wasting half a day on trial and error, you’ll love our newsletter. Get 3 quick reads a week, packed with hard-won lab wisdom. Join FREE here.
Put this article into practice
Choose a free resource to help you move forward
DOWNLOAD
Blood Collection Tube Chart
EBOOK
Guide to Lab Safety











