Buffers are often taken for granted, but they can make or break an experiment. In previous posts, we’ve talked about the wide ranges of buffers available for biological research and the characteristics of a “Good” buffer.
Organic buffers are not inert! They can interact with your experimental molecule, or change pH due to changes in temperature and ion concentrations. So how do you find the right buffer for your experiment? And how can you make sure you’re going to maintain proper pH, even in the face of changing temperatures, ionic strengths, and other variables?
One must choose wisely in picking the right buffer. Below are several factors to consider in finding your perfect buffer mate.
Get in the right pH range
First, determine the optimal pH of your testing environment, and pick a buffer with a pKa value within one pH unit of your test. While more than 20 buffers are commonly used, few have overlapping optimal pH ranges, and none of them have an optimal pH range much greater than 2 pH units. PIPPS, for example, has a range from about 6.9 to 9.2, while CHES has a range from about 8.6 to 10.0.
Put this article into practice
Choose a free resource to help you move forward
DOWNLOAD
Blood Collection Tube Chart
POSTER
Antibiotics Reference Guide
Looking at protein activity? Remember that enzymes only work in very limited pH ranges and organic buffers can inhibit or encourage enzyme functions. You can start by trying a wider-ranging buffer like PIPPS, TAPS or CHES and then use a narrower-range buffer to rule out any buffer effects on your experiment.
Choose a buffer based on your protocol
Your specific experimental methods can determine what type of buffer to use. For example, isolation or purification of proteins can produce false-positive results if the wrong buffer is used.
When considering buffers for ion-exchange chromatography, cation buffers (like Tris) can be used in anion-exchange chromatography, while MES or phosphate, which are anion buffers, are better for cation exchange chromatography. Some Good buffers that contain a piperazine ring (like HEPES, or PIPES) can form radicals, and therefore aren’t very helpful for experiments that delve into oxidation/reduction reactions.
Set the pH value
Buffers are simply an acid and a conjugated base. When making your buffer, you’ll need to get the right concentration of buffer in your experiment and calibrate the pH value for your experiment. Usually done with NaOH/KOH or HCl, calibration involves stirring as you slowly add your acid or base. What’s great now is that there are tables that show you the correct amount of acid/salt and base/salt necessary for any given pH—just look up the amount for your pH and add it!
Now, concentrate
Usually, a good target buffer concentration should be between 10 and 25 mM. However, effective buffer capacity may only be possible at concentrations above 25 mM. At the same time, you don’t want concentrations and ionic strengths that are too high because that combination can inhibit protein function. If your experimental pH changes by >0.05 pH units after adding your protein, then start increasing your concentration of buffer (Good buffers can work up to 50 mM).
Check your temperature
Do you think you’ll be experimenting at physiological temperatures (37°C)? Then look for a buffer that will allow operations at that temperature. Working at 0°C? You’ll probably need a different buffer. Will your experimental temperatures fluctuate? Then you’ll need a buffer that’s not as sensitive to those changes. Tris, for example, is very sensitive to temperature changes altering the pH of the buffer (and therefore, your experiment).
What’s in your water?
It’s almost tragic—you spend good money on high-quality stock buffers that are manufactured to prevent contamination, but then you pour in water from your lab tap. Water that sits in plumbing pipes can attract contaminants and dissolved gases can wreak havoc on your experiment. One solution—just let the water run a bit before adding any to your experiment.
What’s the best buffer, then, will depend a lot on what you’re looking at, and how you’re manipulating your experiment! The following sites can help you calibrate your pH and find the best match for your work:
https://chemwiki.ucdavis.edu/Physical_Chemistry/Acids_and_Bases/Buffers
https://www.science.smith.edu/departments/Biochem/Biochem_353/Common_Buffers.html
https://www.reachdevices.com/Protein/BiologicalBuffers.html
https://www.biomol.net/en/tools/buffercalculator.htm
You made it to the end—nice work! If you’re the kind of scientist who likes figuring things out without wasting half a day on trial and error, you’ll love our newsletter. Get 3 quick reads a week, packed with hard-won lab wisdom. Join FREE here.
Put this article into practice
Choose a free resource to help you move forward
DOWNLOAD
Blood Collection Tube Chart
DIGITAL TOOL
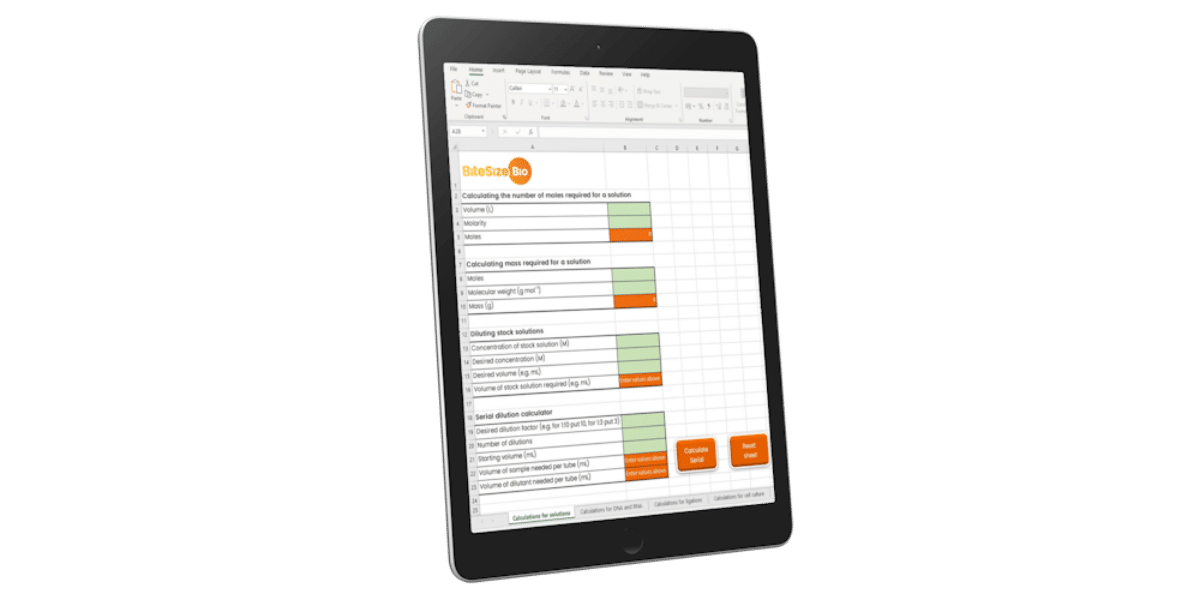
Lab Math Calculator