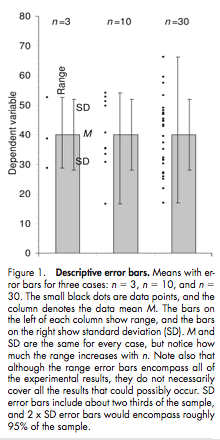
Now that we’ve learned about the role of apoptosis in good health and disease, it will be useful to know how we can detect apoptosis in cells or organisms. A variety of apoptosis detection kits are commercially available, and here is a roundup of how they work:
TUNEL and DNA damage assays
The TUNEL assay is probably the most widely used method to visualize apoptosis in fixed cells. Published by Gavrieli et al. 1992, it relies on the visualization of DNA degradation, more specifically, the identification of nicked DNA by terminal deoxynucleotidyl transferase. This enzyme recognizes broken DNA and inserts dUTPs, which can be tagged a priori and later detected by antibody staining.
This assay can be somewhat insensitive because it depends on DNA damage rather than staining actual components of the apoptotic pathway. Modern assays use other indicators of DNA damage such as antibody staining of phosphorylated histone H2AX, which becomes phosphorylated in the presence of double-stand breaks.
Caspase detection assays
During cell death pathways, a variety of caspases are activated, which in turn cleave molecules via a specific recognition site. Caspase detection assays can be used in vitro or in vivo and exist for initiator or effector caspases in many combinations. Caspase activation assays consist of small inhibitor peptides conjugated to a fluorophore, which are able to specifically target the active site of the chosen caspase. The peptides are cell permeable and can thus also be injected into the circulation of an animal and used to label active caspases in vivo.
Enjoying this article? Get hard-won lab wisdom like this delivered to your inbox 3x a week.

Join over 65,000 fellow researchers saving time, reducing stress, and seeing their experiments succeed. Unsubscribe anytime.
Next issue goes out tomorrow; don’t miss it.
Mitochondrial permeability assays
We learned in previous articles that mitochondria play a pivotal role in cell death, and organelles, including mitochondria, usually disintegrate during apoptosis. Mitochondrial permeability or membrane potential assays make use of a dye that accumulates in healthy mitochondria and aggregates there as a multimer. Upon disruption of the mitochondrial membrane, the dye is released, changing colour due to monomerization as it spreads through the cytoplasm.
Annexin V detection assay
This non-invasive, in vivo method has been developed for apoptosis tracking in humans, specifically in cancer patients, to monitor the effectiveness of treatment. This relies on Annexin V, a protein that can detect phospholipid phosphatidylserine (PS) on the surface of cells marked for apoptosis/phagocytosis by T-cells. In healthy cells, PS is located on the cytosolic side of the plasma membrane and relocates to the outside in the early stages of apoptosis. Radiolabelled Annexin V is used in human patients, while fluorophore- or biotin-tagged AnnexinV is used in non-human assays (Blankenberg 2008).
In vivo detection by fusion proteins
Several protocols have been developed for in vivo imaging of apoptosis activity. They usually consist of a genetically encoded fusion protein comprising a fluorescent protein and a quencher protein, connected by a caspase cleavage signal. Without active caspases, the fluorophore signal is quenched by the coupled protein, but upon caspase activation, the quencher is cleaved off and the fluorophore emits a signal that can be followed by in vivo imaging. Similar results can be obtained by using fluorophores that change color depending on their proximity. Examples are SCAT3 and Apoliner for Drosophila melanogaster (Miura 2011).
Life/death stains
Life/death assays simply use a membrane-impermeable dye that is actively excluded from living cells and thus only accumulates in cells that are dead or dying. These stains, such as DRAQ7, are non-toxic in cell culture and can be used as a long-term indicator. However, they do not discriminate between cells undergoing apoptosis and those dying through other pathways.
Cytotoxicity test kits
These kits are handy as they usually include a combination of the previously described methods. They measure markers of tissue damage such as the release of LDH (lactate dehydrogenase), include a life/death stain and a fluorescent caspase inhibitor. Due to a combination of markers, cells can then be sorted into healthy, early apoptotic, apoptotic or necrotic depending on the state of their decay.
There are a variety of ways to analyze apoptosis and many scientists will choose two or more methods to confirm their research. This review should help make the choice easier for you!
References:
- Blankenberg GD. In Vivo Detection of Apoptosis. (2008) J Nucl Med. 49:81S–95S.
- Gavrieli, Sherman Y, Ben-Sasson SA. (1992) Identification of programmed cell death in situ via specific labelling of nuclear DNA fragmentation. J Cell Biol. 119:493–501.
- Miura M. (2011) Active participation of cell death in development and organismal homeostasis. Dev Growth Differ. 53:125–36.
You made it to the end—nice work! If you’re the kind of scientist who likes figuring things out without wasting half a day on trial and error, you’ll love our newsletter. Get 3 quick reads a week, packed with hard-won lab wisdom. Join FREE here.