Ah, cell counting — it’s the oldest trick in the book! Well, not really, but people have been developing methods for counting cells since the late 1800s. It has been around for a while. But what different methodologies are available to biologists now?
Well, hold on, because you’re in for a treat! In this article, we break down the 4 main types of cytometers: imaging, flow, cell sorters, and time-lapse.
Imaging Cytometer
What Is an Imaging Cytometer?
As the name implies, imaging cytometers statically image a large number of cells and, then, process the images. The resulting read-out provides information on cell quantity and potential cell phenotype. This has been reliably used since the 1950s as an alternative to manually counting cells using a hemocytometer and a light microscope.
How Does Imaging Cytometry Work?
Cells set up for imaging cytometry are either stained to enhance contrast or are stained with antibodies directly conjugated to fluorophores. Cells are then loaded onto a slide and imaged using an imaging cytometer, which analyzes fluorescent intensities as well as identifies individual cells. One of the biggest differences between an imaging cytometer and a flow cytometer is that the former images cells as they cross in front of a light source, whereas the latter uses a photomultiplier to read fluorescence.
Put this article into practice
Choose a free resource to help you move forward
DOWNLOAD
Blood Collection Tube Chart
DIGITAL TOOL
Lab Math Calculator
However, biological tools are always moving forward, and imaging cytometers are no different! Advances in the digital imaging field over the past 30 years have resulted in the development of automated imaging cytometers that can range from cell counters to high-content screening systems. Moreover, some companies offer instruments that function much more like a flow cytometer. The cells are actually introduced via flow stream (i.e., sheath fluid) rather than statically on a slide.
When Should I Use an Imaging Cytometer?
If you are particularly interested in ensuring an accurate cell count on a specific subset of cells, imaging cytometry is for you. This is especially true if you are interested in knowing the diverse distribution of your label within your cells. Flow cytometry cannot detect the distribution of your label within your cells.
Flow Cytometer
What Is a Flow Cytometer?
While they may not be the oldest or the newest cell counters, flow cytometers are the bread and butter of immunology. These machines provide the user with information regarding the phenotype and functional characteristics of various cell populations.
How Does Flow Cytometry Work?
We have previously gone into great detail regarding flow cytometry basics, but it’s your lucky day, Reader, because I love to talk about this topic!
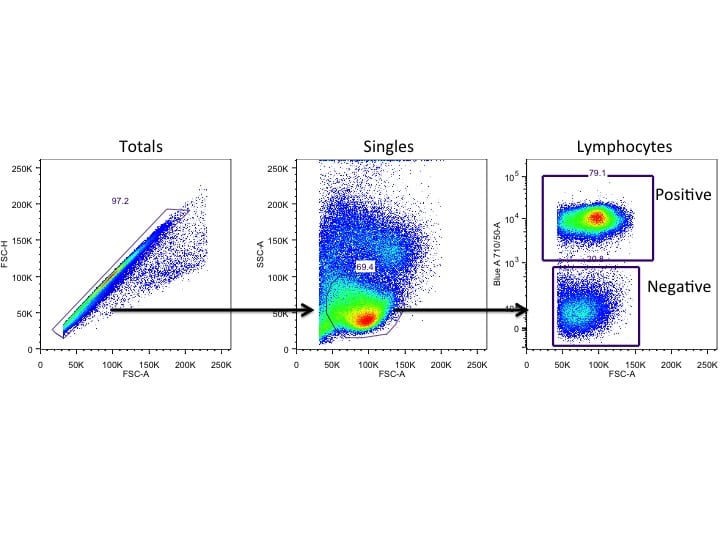
Flow cytometers work when three separate systems come together: fluidics, optics, and electronics. First, cells stained with antibodies directly conjugated to fluorophores are transported via a stream of saline solution to a laser beam (optics). Lasers within the flow cytometer excite these fluorophores at specific wavelengths, which then emit light at specific wavelengths according to the fluorophore of interest. At this point, detectors within the instrument read these emitted light signals and translate them into electronic pulses that are interpreted by a computer. And voila! Raw flow cytometry data.
When Should I Use Flow Cytometry?
If you are interested in really picking apart the phenotype of a population of cells, you should probably use flow cytometry. Many machines now allow up to 18 different fluorophores in a single staining panel! It is pretty obvious that flow and imaging cytometers are incredibly similar, so it really comes down to what you are trying to get out at the end. Flow cytometers cannot pick up on the inhomogenous protein expression between individual cells like imaging cytometers can, but they do have significantly higher throughput.
Cell Sorter
What Is a Cell Sorter?
Cell sorters are flow cytometers capable of sorting cells according to their physical characteristics. Short and sweet.
How Do Cell Sorters Work?
Much like flow cytometry, cells that are destined for a cell sorter are stained with fluorescently-labeled antibodies. You then gate on your populations of interest that you would like to separate. The machine takes care of the rest…following a lot of set-up, of course. The fluid stream that carries cells into the machine is broken up into droplets by a mechanical vibration. These droplets are then electrically charged according the characteristics that you have set as parameters of the cell contained within the droplet. Depending on their charge, the droplets are finally deflected by an electric field into different containers.
When should I Use a Cell Sorter?
A cell sorter is a really cool and useful tool, especially if you want to actually do something with a pure population of cells and not just have phenotypic data (FCM).
Time-Lapse Cytometer
What Is a Time-Lapse Cytometer?
Time-lapse cytometers combine time-lapse microscopy and image cytometry to understand cellular dynamics.
How Do Time-Lapse Cytometers Work?
In essence, time-lapse cytometers are imaging cytometers that monitor cells using time-lapse video recordings. The video is then processed to track cytometric parameters over time.
Time-lapse cytometry is extremely powerful, in part because of two characteristics. First, these machines visually monitor cell morphology or state independent of the use of fluorophores. Thus, cellular damage that sometimes occurs following excitation of fluorophores is a distant memory. Second, time-lapse cytometers do not use heat-generating light sources. Therefore, you can use them in incubators for long-term analysis of your cells without worrying about heat build-up inside the incubator.
When Should I Use a Time-Lapse Cytometer?
As one website so eloquently points out “the visual information can answer questions like — Did the cells really shrivel up or did they die in an apoptotic explosion?” I couldn’t have put it better myself. If you are curious how cells actually behave under certain conditions, for either short or long periods of time, then time-lapse cytometry is your new BFF.
And so concludes our round-up of the 4 types of most commonly used cytometers. I sincerely hope this helped you in navigating your next experimental set-up. Please let me know your favorite method in the comments!
You made it to the end—nice work! If you’re the kind of scientist who likes figuring things out without wasting half a day on trial and error, you’ll love our newsletter. Get 3 quick reads a week, packed with hard-won lab wisdom. Join FREE here.
Put this article into practice
Choose a free resource to help you move forward
DOWNLOAD
Blood Collection Tube Chart
POSTER
Fluorescent Proteins Guide