What’s more disappointing than a failed Western blot?
Several days of work and effort—gone.
After the transfer step, the second biggest cause of failed Western blots is the ECL reagent.
It’s expensive, contains unstable and light-sensitive chemicals, and the stock solution gets abused by careless researchers.
Put this article into practice
Choose a free resource to help you move forward
CHEAT SHEET
SDS-PAGE Protocol Cheat Sheet
CHEAT SHEET
Western Blot Cheat Sheet
So why not save money and make a private stash of ECL?
Skeptical? Well, don’t be. We’re going to learn how to make ECL and explain what its constituent chemicals do. This will improve your Western blot success rate and put you in control if things go wrong.
What Is ECL?
ECL stands for Enhanced Chemiluminescence. As you probably know, we use it to detect and quantify specific proteins on a membrane.
The chemicals in ECL, when mixed together, react to produce light. This reaction is catalyzed by the enzyme horseradish peroxidase.
In a Western blot, the secondary antibody is conjugated to horseradish peroxidase. And because the secondary antibody binds specifically to a primary antibody that binds specifically to a target protein, the emission of light from ECL is proportionate to the amount of that target protein.
What Are the Components of ECL?
Excluding water, ECL has four ingredients. These are:
- Luminol.
- p-Coumaric acid.
- Hydrogen peroxide.
- Tris-HCl at pH 8.5.
As mentioned, these chemicals react to produce light. That is why ECL is supplied as two separate solutions that we mix immediately before the detection step in our Western blot.
We will make our own ECL as two separate solutions, too.
And in case you are wondering, we keep the hydrogen peroxide separate from the luminol and p-coumaric acid, and both solutions are buffered with the tris-HCl.
How Does ECL Work?
Let’s explain this by going through the four ingredients of ECL one by one.
1. Luminol
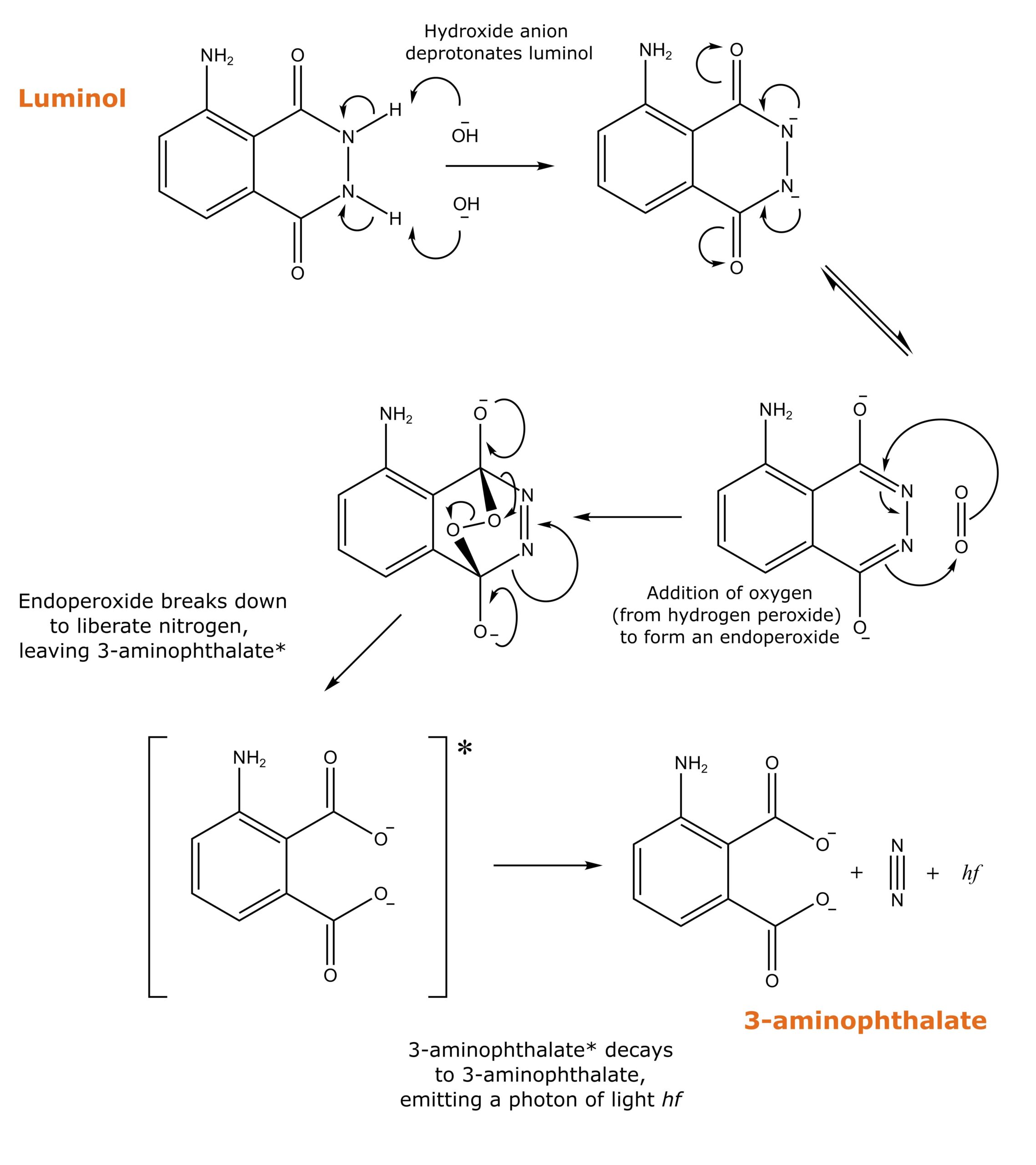
Luminol produces the light needed to detect the target protein. It emits light when it reacts with an oxidizing agent. For the reaction to proceed, luminol needs to be deprotonated. That’s why the ECL reagent is prepared at pH 8.5, which is slightly basic.
The reaction of luminol with an oxidizing agent is complicated and contains many steps. However, we must understand that one product of the reaction is the dianion 3-aminophthalate.
The 3-aminophthalate is produced with an unpaired electron in a non-ground-state orbital. There is no residual electron spin, and this is called the singlet excited state. The singlet excited state is unstable and decays to the ground state by emitting photons.
The wavelength of the photons depends on the solvent in which luminol is dissolved but is usually around 425 nm (blue light).
And when a chemical reacts and produces light, it’s said to be chemiluminescent. Luminol is chemiluminescent.
2. Hydrogen Peroxide
Hydrogen peroxide is an oxidizing agent. It breaks down into water and oxygen, and many species catalyze its decomposition. Examples include silver(I), iron(II), titanium(III), and potassium iodide.
In the ECL reagent, hydrogen peroxide produces the oxygen that reacts with (oxidizes) the luminol in the presence of horseradish peroxidase to produce the chemiluminescence described above.
3. Tris-HCl
Like all buffers, the tris-HCl provides a stable chemical environment for the other components of the ECL reagent.
The slightly basic pH of 8.5 deprotonates the luminol so the chemiluminescent reaction can proceed.
4. p-Coumaric Acid
The chemiluminescent conversion of luminol to 3-aminophthalate is not ideal for immunohistochemistry. The amount of light emitted is relatively low, and it’s all emitted quite quickly, in something akin to a flash.
There are several chemicals, mostly phenol derivatives, that rectify this issue. p-Coumaric acid is one of them.
It increases the light emitted by luminol alone by about three orders of magnitude. And remarkably, it massively extends the emission period without changing the wavelength of the emitted photons. [1] In fact, p-coumaric acid loses only about 70 of its emission intensity after one hour.
How Luminol Emits Light via Chemiluminescence
Figure 1 depicts how luminol reacts with the other components of ECL to emit light via chemiluminescence.
How To Make ECL
So, on to what you came here for. First, check if you have the ingredients in your lab. Then, prepare the following solutions:
- Luminol, 250 mM, in DMSO.
- p-Coumaric acid, 90 mM, in DMSO.
- 1M Tris-HCl pH at 8.5.
Hydrogen peroxide is supplied as a 30% solution in water, so that one is made for you.
Use opaque receptacles or wrap clear ones in aluminum foil to make them opaque. Now, keeping everything cold and dark, prepare two solutions as per Table 1 below.
Table 1. Ingredients to prepare your own ECL reagent.
Ingredient | ECL Solution A | ECL Solution B |
250 mM luminol | 1 mL | - |
90 mM p-coumaric acid | 0.44 mL | - |
1 M tris-HCl pH 8.5 | 10 mL | 10 mL |
30 % Hydrogen peroxide | - | 0.064 mL |
Water | 88.56 mL | 89.936 mL |
There are a few extra points to note.
Store both solutions in the dark at 4 °C. If stored correctly, they should last for one or two months. When your Western blots start looking faint or failing, it’s time to prepare the solutions again.
How To Use Your ECL
So now you’ve learned how to make your own ECL, you might wonder: “do I use it any differently from regular ECL?”
Nope!
Use your homemade ECL identically to the premade stuff. Combine equal volumes of each solution in a separate tube and mix thoroughly. Then, cover your blot with the solution and leave for one minute. Drain off the excess fluid and visualize your blot. Simple.
Why Make Your Own ECL?
Hopefully, the benefits of preparing your own ECL are clear, but in summary:
- It saves money.
- It’s yours.
- You can look after it.
- It won’t unexpectedly run out.
Can I Use Other Enhancers in My ECL?
Yes, you can! There are alternatives to p-coumaric acid. You can check out those discussed in this paper by Thorpe et al. [1]
And more recently, researchers have explored 4-iodophenylboronic acid as an alternative enhancer. [2] The primary benefit of it over p-coumaric acid is lower background chemiluminescence. Have a play around!
Home Brew ECL in Summary
You’ve learned how to make ECL reagent and perhaps learned a little about what the ingredients do.
This information will make you better at Western blots and a better scientist!
If you’ve got any hints to help prepare ECL in the lab, then let us know in the comments section below. The same goes for any helpful Western blot wisdom.
For more tips, tricks, and hacks for getting your experiments done, check out the Bitesize Bio DIY in the Lab Hub.
Want all your Western blot buffer recipes, an ECL reagent recipe, and an easy protocol all in one place and on hand? Download our free Western blot cheat sheet.
Originally published June 2012. Revised and updated November 2022.
References
- Thorpe GH, Kricka LJ, Moseley SB, and Whitehead TP (1985) Phenols as enhancers of the chemiluminescent horseradish peroxidase-luminol-hydrogen peroxide reaction: application in luminescence-monitored enzyme immunoassays. Clin Chem 31:1335–41
- Haan C and Behrmann I (2007) A cost effective non-commercial ECL-solution for Western blot detections yielding strong signals and low background. J Immunol Methods 318:11–9
You made it to the end—nice work! If you’re the kind of scientist who likes figuring things out without wasting half a day on trial and error, you’ll love our newsletter. Get 3 quick reads a week, packed with hard-won lab wisdom. Join FREE here.
Put this article into practice
Choose a free resource to help you move forward
CHEAT SHEET
SDS-PAGE Protocol Cheat Sheet
CHEAT SHEET
Western Blot Cheat Sheet








