Phagocytosis is a fascinating innate immune cellular process in which professional phagocytes—including macrophages, neutrophils, monocytes, and dendritic cells—ingest unwelcome particles > 0.5 µm in diameter.
This cellular “eating” encloses the offending target into an intracellular membrane phagosome, where it is digested and inactivated by a chemical attack. This article provides tips and tricks on live imaging of phagocytosis.
The Problem with Imaging Phagocytosis
Much of my time in the lab was spent at the microscope imaging phagocytes devouring these unwanted particles. Over the years I would hear from students in other labs who were trying to measure phagocytosis.
It was difficult to say whether a target had been internalized or not from their single image because it is tricky to see whether a target is inside, beside, atop, or outside the phagocyte.
Put this article into practice
Choose a free resource to help you move forward
POSTER
Histological Stains Poster
POSTER
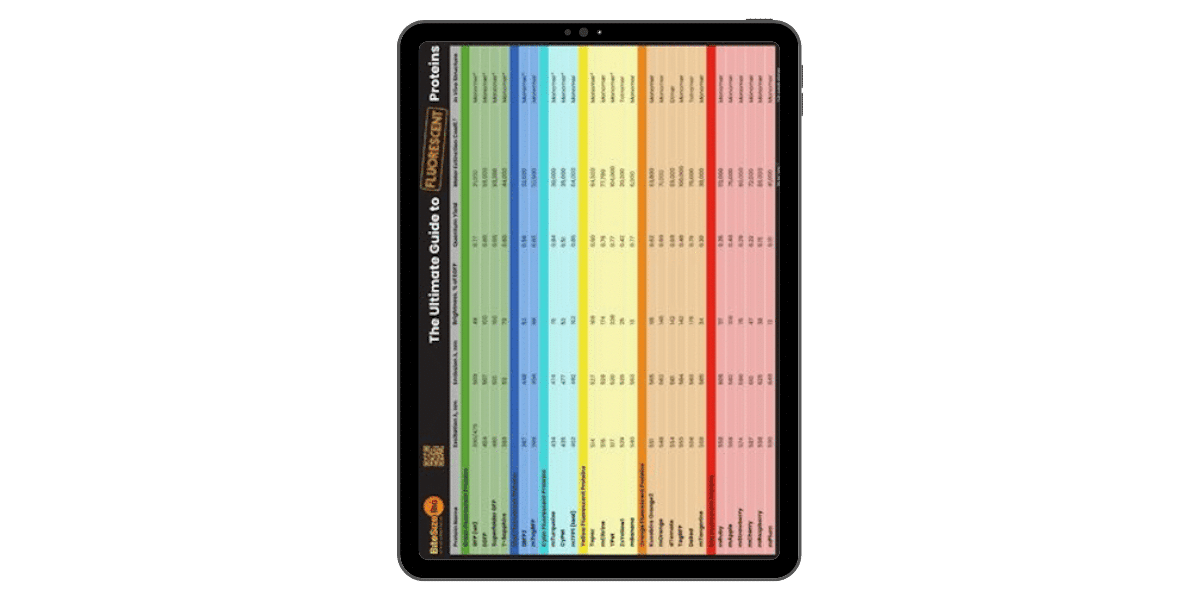
Fluorescent Proteins Guide
Live Imaging of Phagocytosis
Live imaging of phagocytosis, on the other hand, not only gives a clear answer to the in-or-out question, but also reveals the subtleties of phagocytic dynamics, and sometimes the unexpected.
Live imaging recently showed that macrophages can physically fold up lengthy fungal filaments, adding physical damage to the list of intracellular assaults faced by internalized targets. [1]
If you’re keen to capture phagocytosis on camera, we’ll talk you through a few key considerations.
Which Phagocytes to Use
I studied phagocytosis with macrophages—large, adherent “big eaters”, which are relatively easy to handle with standard tissue culture (TC) methods.
Mouse cell lines, such as J774.1 and RAW264.7, can be sourced and grown in standard TC flasks using DMEM or RPMI medium with supplements, in humid incubators at 37ºC with 5% CO2. Fetal bovine serum (FBS) supplement should be heat-inactivated to exclude the effects of complement—important if you study receptor-driven phagocytosis.
If you want to use primary cells, bone marrow-derived macrophages (BMDM) from mice or human monocyte-derived macrophages (HMDM) from blood can be cultured in vitro. In my experience, journal manuscript peer reviewers seem to prefer research that uses primary macrophages, rather than cell lines alone.
When using primary macrophages, be sure to schedule their extraction in alignment with your microscope booking.
Don’t forget to refresh the TC medium to keep your macrophages happy—happy macrophages will adhere to the flask and appear stretchy, not rounded up.
Choosing Phagocytic Targets
Fungi as phagocytic targets were the focus of my research. The size of yeast cells or fungal spores (3–5 µm diameter), or hyphae or mycelia of varying length), give easily visualized particles with differential interference contrast (DIC) alone, compared with smaller targets such as bacteria.
Another popular target for phagocytosis studies is beads, which can be obtained in a variety of sizes and fluorescent colors. Carboxyl-coated beads are more eagerly phagocytosed than uncoated beads.
The multiplicity of infection (MOI) is a ratio of target to phagocyte, and choosing the right MOI matters. For larger targets (e.g. yeast cells) try 3:1 (target: phagocyte) and for smaller targets, try an MOI of e.g. 10:1.
Imaging Dishes that Give Great Results
Choose a confocal-quality glass or plastic imaging dish such as the ibidi 8-well µ-slide. You can image different conditions in wells using multi-point acquisition, and the 1-cm2 area makes for an economical use of your precious cells.
You might consider setting up parallel phagocytosis interactions in a second dish. You can then fix the cells at an appropriate time point, for immunocytochemistry (ICC) staining—all the while using small volumes of ICC reagents.
Cell Density is Important
For live imaging, aim to have the right density of cells—around 80% confluence on the day of the experiment. Try adding 0.5–1.0 × 105 macrophages per 1 cm2 (lifted from culture, counted, and pipetted with fresh medium into the imaging dish), allowing at least 8 hours for macrophages to adhere down.
Microscope Set-up for Live Imaging of Phagocytosis
Essential Conditions for Live Imaging of Phagocytosis
Live imaging of macrophages requires a temperature of 37ºC, maintained with a heated microscope stage or enclosed chamber. CO2 at 5% can be more difficult to deliver during imaging. An alternative is to use CO2-independent medium, which works well for the BMDM and HMDM cell lines mentioned above, but beware that finicky cells may not like it.
Practice the Microscope Set-up Ahead of Time
Before you set up a huge experiment, make sure you know how to set up the inverted microscope with camera and acquisition software. Ask for help from a knowledgeable colleague.
You want to be prepared because spending time figuring out the imaging settings after adding targets to phagocytes, positioning the slide on the microscope, and focusing could mean that much of the phagocytic action has already happened.
Think Ahead to the Desired Outcome
Time-lapse images taken one or two minutes apart will give a reasonably smooth phagocytosis movie when run together at 12 frames per second.
If you’re imaging with multi-point visiting, remember that it may take your microscope stage a few seconds to move between each point. Think about what is feasible for your microscope and the resolution (in time and magnification) of what you are aiming to capture.
If you need finer resolution in time, image fewer locations per time point, but with smaller time intervals. Collect pilot data to get a broad sense of the dynamics of your cell interactions and work with that knowledge to hone parameters to suit your needs.
Ready, Steady, Go
With everything as ready as possible, you can now unleash the targets.
- Remove a portion of the culture medium from the well, taking care not to disturb the adhered phagocytes with your pipette tip.
- Add back the same volume of fresh medium containing the correct number of targets to give the desired MOI.
- Pipette gently to avoid blasting adhered cells away with jet pressure.
If you plan to use fluorescent stains, figure out if they should be used hours ahead to pre-stain cells (such as live actin stain SiR-actin), or if the stain can be added at the last moment (such as LysoTracker™ Red, as used in Lewis et al. [2]). If your stain is very faint and high laser power is needed to see the signal, you might fry your cells.
Data
Data will be generated from every location on the slide imaged, for every channel you image in (DIC and each fluorescent channel), and for every time point you capture. Expect a lot of data from live imaging.
Think ahead about how you will manage data at the point of acquisition, how to transfer data for analysis, and how to store the data securely. You can expect to spend a long time processing and analyzing the data, but you’ll have some pretty amazing movies generated at the end of all that work.
Live Imaging of Phagocytosis Summarized
Live imaging of phagocytosis can feel like you need the alignment of the planets. But with good planning and preparation, all you need is the alignment of phagocytes, targets, and your microscope.
Do you have any other top tips for live imaging phagocytosis? We’d love to hear from you in the comments.
References
1. Lewis LE, et al. Live-cell video microscopy of fungal pathogen phagocytosis. J. Vis. Exp. 2013 (71): e50196. doi:10.3791/50196.
2. Bain JM, et al. Immune cells fold and damage fungal hyphae. PNAS 2021;118 (15): e2020484118. doi:10.1073/pnas.2020484118.
You made it to the end—nice work! If you’re the kind of scientist who likes figuring things out without wasting half a day on trial and error, you’ll love our newsletter. Get 3 quick reads a week, packed with hard-won lab wisdom. Join FREE here.
Put this article into practice
Choose a free resource to help you move forward
POSTER
Histological Stains Poster
POSTER
Fluorescent Proteins Guide