I have worked in flow cytometry for a number of years. I’m still annoyed that many myths and imprecisions are perpetrated and perpetuated. Here is my non-exhaustive list of cytometry-related beliefs that send flow cytometrists screaming from the room or at least, being English, make me tut sadly.
Forward Scatter Equals Cell Size
No No No! The way cells scatter light is a complex affair involving refraction, reflection, and diffraction of the light. It is an optical measurement. By far the biggest influence on forward scatter is the difference between the refractive index of the inside and the outside of the cell. But things like nuclear to cytoplasmic ratio, the number and type of cellular granules and the surface topography of the cells also influence the way cells scatter light. Therefore, don’t always assume a larger forward scatter signal is equal to larger cell size.
Cells were Fixed in 1% Paraformaldehyde
No, they weren’t. Paraformaldehyde (PFA) is an insoluble white powder. Is that what you used? You may have heated paraformaldehyde powder until it dissolved or used some other technique to get it into solution, but once it is in solution it is actually formaldehyde. This was elegantly discussed in a great post to the Purdue cytometry list a few years ago. Unfortunatley the information is often overlooked and inaccurately described.
Cells Were Examined on a FACS
Despite very many explanations, this one still persists. FACS is an acronym; it stands for Fluorescence Activated Cell Sorter. You probably didn’t use a sorter but an analyzer so that’s error 1. Error 2 is that FACS is a tradename of Becton Dickinson so while all FACS machines are flow cytometers, not all cytometers are FACS machines. It’s like calling all vacuum cleaners ‘Hoovers,’ or all ballpoint pens ‘Biros.’ Let’s just talk about flow cytometry and doing your experiments on a flow cytometer.
Put this article into practice
Choose a free resource to help you move forward
DOWNLOAD
Blood Collection Tube Chart
CHECKLIST
Journal Club Checklist
I Used a Red Fluorochrome
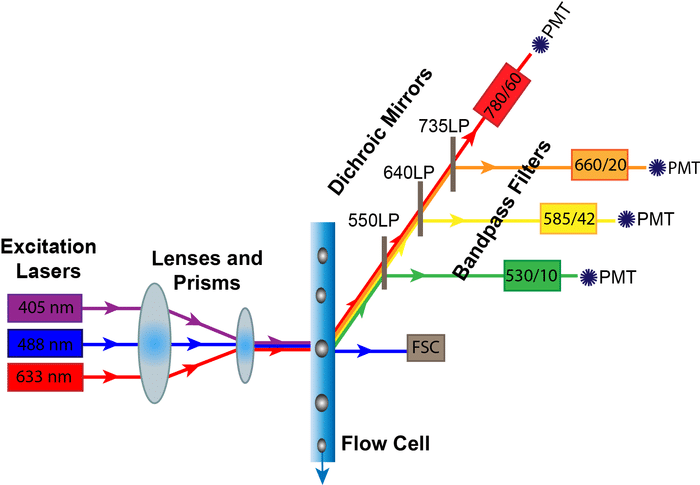
OK, the cells looked red under the microscope but we, as cytometrists, know that ‘red’ can mean anywhere on the electromagnetic spectrum from around 640nm to 800nm. APC, PE-Cy7, Red Fluorescent Protein are all ‘red’ but they have very different excitation and emission spectra. This is especially annoying to those of us who run cell sorters as it often requires a lot of probing questions till we get the right information (i.e., the excitation and emission spectra of the dyes), so we can set the appropriate excitation wavelength and optical filters in the machine.
I Measured Fluorescence in the FL1 Channel
Now, way back in the day, we only had one or two cytometers that measured only one or two colors. Then, I was pretty certain that FL1 was a detection channel for a green fluorochrome (see point 4 above, I am falling into the trap myself!) but now I am not so sure. In some cytometers, the FL1 channel is the first one measured on the spectrum, which may well be a violet or ultraviolet detection channel. I can almost forgive people saying this, but can’t forgive seeing it in print. We recently blogged on this channel about MIFlowCyt guidelines. The guidelines recommend labeling axes at a minimum with the analyte, fluorochrome and optical filter. This is all the information I need to know what the researcher used.
It’s been quite a feat to whittle this list down to five, but that’s enough for now. I need a lie down but while I do so, why not tell us about your favorite irritations in the comments section below.
You made it to the end—nice work! If you’re the kind of scientist who likes figuring things out without wasting half a day on trial and error, you’ll love our newsletter. Get 3 quick reads a week, packed with hard-won lab wisdom. Join FREE here.
Put this article into practice
Choose a free resource to help you move forward
DOWNLOAD
Blood Collection Tube Chart
CHEAT SHEET
Guide to Pipette Cleaning