Wow, you’ve done it! Your experiment worked and your boss asked you to write it up for publication in your favorite journal. Where to start with presenting your flow data? Take a deep breath, help is in hand in the form of the MIFloCyt Guidelines.
As with other scientific techniques, there are ‘Minimum Information about a Flow Cytometry Experiment (MIFlowCyt)’ guidelines and these guidelines are now being used my several journals including Cytometry Part A, PLOS and all Nature publications.
In 1960, the invention of the LASER was reported in less than 300 words. However, the majority of scientific research requires more information, diagrams, figures, images, and data sets than that. Using the MIFlowCyt guideline, this process is easy as it gives step-by-step guidelines to follow. Don’t worry you can use the supplementary information to fulfill all of the guidelines.
Here are a couple of the MIFlowCyt guidelines…
Experimental overview: Tell the reader why you did your experiment, what was your hypothesis? What variables were in the experiment, what QC did you carry out? What are the conclusions of your study?
Enjoying this article? Get hard-won lab wisdom like this delivered to your inbox 3x a week.

Join over 65,000 fellow researchers saving time, reducing stress, and seeing their experiments succeed. Unsubscribe anytime.
Next issue goes out tomorrow; don’t miss it.
Samples: What samples were used, from what source? List any treatments used. A really important thing to list is the reagents used, such as the analyte, the analyte detector and the reporter. For example, if you are using looking at the cell surface protein CD4, then this is the analyte. The analyte detector will be your anti-CD4 antibody that specifically binds to the analyte. If your CD4 is bound to PE, then this fluorochrome would be the reporter. A table is a great way to state this information. Don’t forget to add the company you purchased the reagents from and list any antibody clones. Remember you are giving the details so that someone could pick up your paper and easily repeat it.
Instrument details: You might not realise it but the cytometer you did your experiment could influence your results. Especially as cytometers vary in laser power, laser excitation wavelengths, lenses, mirrors, filters, and detectors. Therefore, it is imperative that you list the type of cytometer, lasers, and filters used in an experiment.
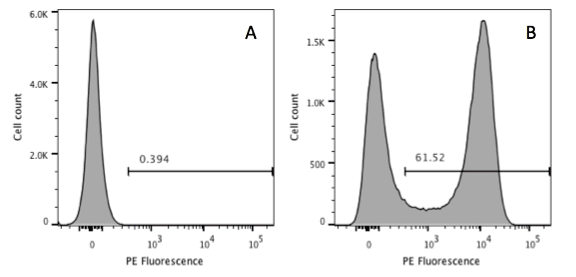
Data analysis: Cytometers produce list-mode data files in the form of .FCS files. These files contain your raw data as well as some metadata. Some journals are now asking for your raw data to be deposited in repositories (for example https://flowrepository.org/). An example of your compensation matrix should be added to the supplementary data so that readers can evaluate your data in context of your (hopefully) correct compensation. Describing how you gated your samples is really important, so write whether the gating was set due to FMO controls or based on a negative sample etc. You will need to add some descriptive statistics as well, such as percentage positive or MFI (don’t forget to say if it’s a Mean Fluorescence Intensity or Median FI). An example gating scheme should be included in your supplementary information.
It might seem like a lot of work, but it’s all the information that is needed to repeat your experiment and it’s better to add it in first before a reviewer asks you to rewrite your manuscript to include it. More information on MiFloCyt guidelines can be found at https://isac-net.org/Resources-for-Cytometrists/Data-Standards/MIFlowCyt.aspx.
You made it to the end—nice work! If you’re the kind of scientist who likes figuring things out without wasting half a day on trial and error, you’ll love our newsletter. Get 3 quick reads a week, packed with hard-won lab wisdom. Join FREE here.