If you use ultraviolet (UV) light in the lab, have you ever stopped to consider how UV light damages DNA and causes mutations? If not, we’ve got the lowdown on how to protect yourself—and your experiments—right here.
How UV Light Damages DNA
Ultraviolet (UV) radiation is a carcinogen because it damages DNA, resulting in mutations in proto-oncogenes and tumor suppressor genes. [1–3] The connection between overexposure to sunlight—and more specifically to the UV portion of its spectrum—and cancer is well documented. [4,5]
This is why you should always follow the advice—summed up so eloquently by Baz Lurhmann—to apply sunscreen to protect yourself from skin cancer. Exposure to UV radiation is also associated with photoaging and damage to the eyes. The extent and type of DNA damage largely depend on the wavelength of the incident photons. [1]
Learning how UV radiation causes DNA mutations is key to gaining a solid understanding of the mechanisms underlying skin cancer. This is also important when considering the detrimental effects that UV light can have on your DNA-based experiments. In cells, DNA repair mechanisms can fix UV-damaged bases, but in purified plasmids, no such mechanisms exist, and unrepaired UV damage can be detrimental to the success of downstream applications.
In this article, we will:
- highlight the two main mechanisms by which UV light can cause mutations;
- highlight lab equipment that uses UV light and the types of UV light involved;
- explain why this is important when working with DNA in the lab;
- provide tips for working safely with UV light sources—both for your samples and yourself.
What is UV Radiation?
UV radiation is the part of the electromagnetic spectrum that lies between 200 and 400 nm, with shorter wavelengths than violet of the visible spectrum (hence the name, ultraviolet). This range is further divided into short wave (200–280 nm, UV-C), middle wave (280–320 nm, UV-B), and long wave (320–400 nm, UV-A) light. [1]
Our eyes can’t directly perceive UV light because the lens of the human eye blocks most radiation between 300 nm and 400 nm, while the cornea blocks shorter wavelengths of UV light (i.e., UV-B and UV-C).
UV-C: Damaging Reality for DNA
The shorter wavelength UV-C light carries with it significantly more energy than its long wave UV-A counterpart, and is much more damaging to DNA. This increased capacity for DNA damage of UV-C is also because DNA absorbs light at a maximum of 260 nm, which is within the UV-C range. [1]
Thankfully, though, solar UV-C rays do not reach the earth because they are absorbed by the ozone layer. [1] However, as we discuss later on in this article, UV-C is used in the lab and can, therefore, inflict DNA damage if proper safety precautions are not taken.
UV-B and UV-A: Still Dangerous
While UV-C poses the biggest danger, UV-A and UV-B rays are not harmless. UV-B and UV-A rays are two forms of high-energy radiation that ionize (i.e., remove electrons from) molecules in a photochemical reaction that generates new molecular products. UV-B and UV-A rays can penetrate the epidermis and dermis layers of our skin, respectively, and have been categorized as “Class I carcinogens” by the International Agency for Research on Cancer (IARC). [6] Importantly, while UV-A light is less energetic than UV-B, it is 20 times more intense, but this ratio varies depending on latitude and season. [1]
Moreover, UV-B-induced DNA damage has been associated with the onset of non-melanoma skin cancers (NMSCs), which include basal cell carcinoma (BSC) and squamous cell carcinoma (SCC). To make matters worse, damage to the stratospheric ozone layer has led to an increase in UV-B radiation on earth, along with a parallel increase in the incidence of NMSCs and malignant melanomas. [7] Even more reason to slather on that sunscreen, folks!
Main Types of UV-Induced Mutations
The efficient repair of DNA damage prior to replication is essential for preventing carcinogenesis. If this doesn’t happen or if DNA damaged cells are not eliminated by apoptosis, DNA lesions in damaged cells express their mutagenic properties in a process that activates or inactivates proto-oncogenic or tumor-suppressing genes, respectively. [1]
UV damage occurs via two distinct types of mutations: dimerizing and oxidative. Let’s discuss these in more detail.
1. Dimerizing Mutations
Exposure to UV-B, and to a lesser extent UV-A, rays leads to the formation of the most common photochemical products in DNA—cyclobutane pyrimidine dimers (CPDs), as well as pyrimidine (6–4) pyrimidone photoproducts (64PPs). CPDs form when two adjacent pyrimidines (thymines, TT, or cytosines, CC) become covalently linked by their C=C double bonds. These four carbons form a cyclic ring (cyclobutane) that links the two pyrimidines, thus creating a chemical intermediate that is not normally found in DNA. This photochemical product causes a structural kink in the DNA that prevents the pyrimidines from base pairing, and prevents DNA replication. Unlike oxidative mutations, UV-induced dimerization mutations arise from the direct absorption of UV photons (Figure 1). [1]
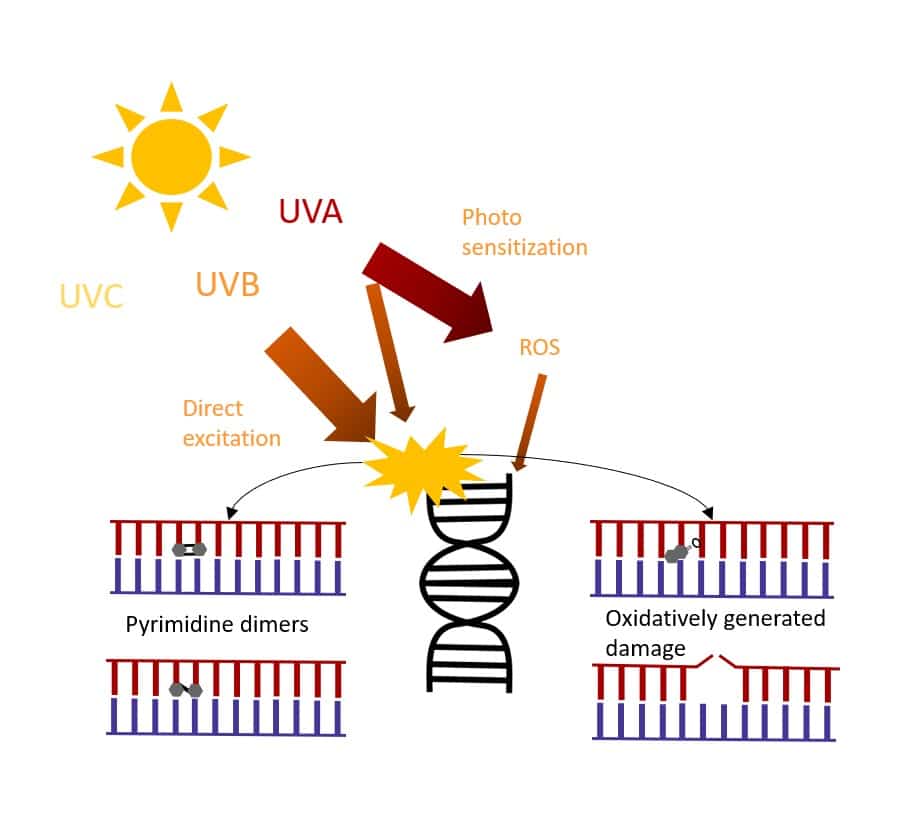
Figure 1. Types of UV-induced DNA damage caused by UV-A and UV-B light.
2. Oxidative Mutations
UV exposure doesn’t always lead directly to mutations in the DNA. In fact, UV-A radiation commonly damages DNA in an oxygen-dependent manner that involves photosensitization. This leads to the production of a free radical that then interacts with and oxidizes DNA bases. These oxidized bases don’t pair correctly during replication, resulting in mutations (Figure 1).
One example of this is a G to T transversion mediated by reactive oxygen species (ROS). The oxidation of guanine into 8-oxoguanine prevents the hydrogen bonding required to base pair with cytosine. Instead, during replication, 8-oxoguanine can base pair with adenine via two hydrogen bonds. When the second strand is synthesized, the base position originally occupied by a guanine is then replaced with a thymine, leading to a G to T transversion. Oxidative mutations can also result in single-stranded breaks. [1]
What Lab Equipment Uses a UV Light Source?
While UV light induces DNA damage and burns, making it potentially dangerous, it is a double-edged sword in that it’s also very useful in biological research. Therefore, solar UV is not the only concern for UV-induced DNA damage and skin cancer. Tanning beds, as well as several types of lab equipment, also utilize UV light. Common pieces of lab equipment that utilize a UV light source include transilluminators, biological safety cabinets, and UV crosslinkers (Table 2). In some cases, these devices can emit intense UV radiation that is more concentrated, and, therefore, more damaging to DNA than natural UV radiation from the sun. [1]
Why is This Important for Your Safety in the Lab?
Some pieces of lab equipment emit high-intensity UV radiation. This is hazardous because direct exposure to the skin and eyes can produce visible and painful burns as well as invisible DNA damage. For your own safety, it is important to be aware of the dangers of UV light and the precautions you can take to keep yourself and others safe in the lab. This is also important so you can make sure that your DNA and cell samples are not ruined by exposure to UV light.
Dangers of UV Light Exposure for the Eyes
Eyes are generally sensitive organs and this is no different when it comes to UV exposure. The epithelial cells of the cornea absorb light in the actinic range of the UV spectrum (200–320 nm), and exposure to this can cause symptoms of photokeratitis, which do not manifest for several hours after the initial exposure.
Symptoms of photokeratitis include sensitivity to bright light, watery eyes, and the feeling of having sand in the eye. The effects usually go away after 48 hours and continue to disappear as the cells of the cornea are replaced. However, there is also a risk of long-term effects.
While the majority of UV radiation that enters the eye is absorbed by the cornea, UV-A is absorbed by the lens of the eye, which can lead to protein damage in the lens and subsequent formation of a cataract.
Dangers of UV Light Exposure for the Skin
Excessive exposure of the skin to UV in the actinic range (200–320 nm) results in symptoms that are similar to sunburn, including redness, swelling, pain, blistering, and peeling skin. Not pleasant!
A number of factors can also affect the degree of skin damage from UV light exposure. These include use of drugs (e.g., tetracycline), the degree of melanin in the skin, and photosensitization from certain foods (e.g., figs, limes, parsnips, and celery root).
Short-term skin damage often heals quickly, but chronic exposure to UV may increase your risk of developing skin cancer.
How Can You Protect Yourself from UV Light Exposure in the Lab?
Examples of lab equipment that uses different types of UV radiation are detailed in Table 1, along with some safety precautions for their use and UV alternatives you can use in the lab.
Table 1. Examples of lab equipment that uses UV radiation.
Lab equipment and UV light sources | Purpose of equipment | Safety precautions for you | Alternatives to UV |
Transilluminator | To visualize DNA bands in agarose gels | Always wear a visor, gloves, and a lab coat – and button it up to the top! Make sure you use the transilluminator in an isolated room so others are not inadvertently exposed There should be some warning system to indicate its use (e.g., red warning light outside the room in which the transilluminator is being used) | Non-UV box or dye visible in natural light (e.g. crystal violet) |
Gel imaging systems | To image different types of gels (e.g., agarose gels and SDS-PAGE gels) | Make sure the door of the imaging system is fully closed before imaging and exposure of the gel to UV light | Fluorescent (e.g. Pro-Q™ Diamond Phosphoprotein Stain) or colorimetric (e.g. SimplyBlue™ Safe Stain, InstantBlue™, Coomassie blue or silver stain) protein stains. Keep in mind that Coomassie blue and silver staining reagents need to be disposed of in hazardous waste |
UV crosslinker | To crosslink nucleic acids to other macromolecules and/or surfaces | Although most crosslinkers are closed systems, you should check that there are no “light leaks” Always wear a visor, gloves, and a lab coat | Chemical or synthetic bioconjugation (e.g. on-support conjugation, solution-phase conjugation) |
UV lamps in biological safety cabinets | UV-C radiation penetrates the cell membrane of living cells and disrupts the DNA molecules, preventing cell replication and helping to decontaminate the surfaces of biological safety cabinets However, surfaces should always be cleaned with an additional decontaminating agent and not with UV alone. This is because UV light can only kill microorganisms that it can reach. If, for example, there is dust on the UV bulb, if microorganisms are present on uneven surfaces (e.g. between the seams of biological safety cabinets), or if they are covered by a layer of dust, these microorganisms will be protected from UV-induced damage | Ensure that while the UV cycle is on, the sash on the cabinet is completely down and you do not leave your samples in the cabinet during this time Once the UV cycle is complete, switch the UV lamp off and make sure it stays off while you are working in the biological safety cabinet | Spray with 70% (v/v) ethanol or isopropanol |
Why Is It Important to Protect Your DNA Samples from UV Radiation in the Lab?
Well, a plasmid that contains UV-induced dimerizing mutations is unlikely to be replicated efficiently in E. coli. The structural change brought about by dimerizing mutations leaves the plasmid DNA available to repair enzymes. However, errors in repair commonly lead to the replacement of cytosine for thymine, thus changing the original DNA sequence in potentially detrimental ways that can affect your downstream experiments.
When cutting out purified plasmids, DNA fragments, or PCR products from an agarose gel, it’s important to be quick! Don’t spend 30 minutes with your gel on the transilluminator while you carefully and skillfully cut out the relevant bands. Quickly identify and excise the bands you need to limit the introduction of mutations in your DNA samples. And just to be sure, sequence everything after purification! This can save you a lot of heartache down the line.
To go one step further, you could also use non-UV boxes to visualize DNA bands in your agarose gel. Alternatively, there are now gel purification kits that allow users to go “UV free”. For example, the S.N.A.P.™ UV-Free Gel Purification Kit uses a crystal violet reagent that allows for the visualization of DNA in an agarose gel under ambient light.
This also eliminates the need to use ethidium bromide or other potentially questionable nucleic acid dyes.
Summary of How UV Light Damages DNA
Both long and short wavelength UV light is damaging to DNA, but in different ways. Short wavelength UV-B and UV-C light can directly cause dimerization of pyrimidines, directly prevent replication of plasmid DNA, or induce mutations after faulty repair.
Long wavelength UV-A light is generally less directly damaging, and instead causes mutations through the production of reactive oxygen species. In the lab, UV-A is less harmful to naked DNA. This is why it is best to use a long-wavelength transilluminator to visualize DNA bands, if possible. However, with enough exposure, UV-A light could still damage your DNA.
Hopefully, this article has taught you not only the importance of using a broad-spectrum sunscreen, but also the chemistry behind the damaging effects of UV light.
What other questions do you have about UV light or DNA mutations? Let us know in the comments.
UV light isn’t the only danger to your DNA samples in the lab, read our article on 5 ways to damage DNA to discover other things to avoid.
FAQs
Q: How effective are the DNA Repair mechanisms in different types of cells?
A: DNA repair mechanisms are sophisticated and vary widely among different types of cells and organisms.
In humans, there are several pathways dedicated to repairing UV-induced DNA damage.
The nucleotide excision repair (NER) pathway is critical for fixing UV-caused lesions such as cyclobutane pyrimidine dimers (CPDs) and 6-4 photoproducts. [8]
This pathway excises a small segment of the single-stranded DNA around the damage, allowing DNA polymerase to synthesize a correct patch using the undamaged strand as a template.
In simpler organisms like bacteria, the repair mechanisms might not be as complex, but they often include a form of NER or other repair processes such as photo-reactivation. [9]
Photo-reactivation involves the enzyme photolyase, which directly reverses the damage caused by UV light, specifically CPDs, using energy from visible light.
Q: What are the specific health risks associated with long-term exposure to UV-A and UV-B in a laboratory setting?
A: The health risks associated with long-term exposure to UV-A and UV-B in laboratory settings extend beyond the immediate effects like skin burns or photokeratitis.
Prolonged exposure can lead to serious conditions such as melanoma and other skin cancers, as well as cumulative damage to the eyes that might accelerate cataract development. [10]
Laboratory professionals should adhere strictly to safety protocols to minimize these risks.
Q: How can I efficiently monitor and measure the amount of UV exposure in a laboratory to ensure safety?
A: If you believe UV exposure is an issue in your lab, use UV radiation meters and dosimeters that can provide real-time data on UV light levels to monitor and measure the amount of UV exposure.
These devices help ensure UV radiation does not exceed safe thresholds, enabling you or your lab manager to implement effective safety measures.
Regular audits and maintenance of UV-emitting equipment also help to keep exposure within safe limits and safeguard the health of lab personnel.
References
- Cadet J, Douki T. (2018) Formation of UV-induced DNA damage contributing to skin cancer development. Photochem Photobiol Sci 17(12):1816–41.
- Sinha RP, Häder D-P. (2002) UV-induced DNA damage and repair: a review. Photochem Photobiol Sci 1(4):225–36.
- Friedberg EC, Walker GC, Siede W, Wolfram S. (1995) DNA Repair and Mutagenesis. ASM Press.
- Koh HK, Geller AC, Miller DR, Grossbart TA, Lew RA. (1996) Prevention and early detection strategies for melanoma and skin cancer. Current status. Arch Dermatol 132(4):436–43.
- Armstrong BK, Cust AE. (2017) Sun exposure and skin cancer, and the puzzle of cutaneous melanoma: A perspective on Fears et al. Mathematical models of age and ultraviolet effects on the incidence of skin cancer among whites in the United States. American Journal of Epidemiology 1977; 105: 420-427. Cancer Epidemiol 48:147–56.
- El Ghissassi F, Baan R, Straif K, Grosse Y, Secretan B, Bouvard V, et al. (2009) A review of human carcinogens—Part D: radiation. The Lancet Oncology 10(8):751–2.
- Lucas RM, Norval M, Neale RE, Young AR, de Gruijl FR, Takizawa Y, et al. (2015) The consequences for human health of stratospheric ozone depletion in association with other environmental factors. Photochem Photobiol Sci 14(1):53–87.
- Kusakabe M, Onishi Y, Tada H, et al. (2019). Mechanism and regulation of DNA damage recognition in nucleotide excision repair. Genes and Environ 41(2).
- Clancy S. (2008). DNA damage & repair: mechanisms for maintaining DNA integrity. Nature Education 1(1):103
- Moorfields Private Eye Hospital. Looking after your eyes in the sun. URL: https://www.moorfields.nhs.uk/private/about-moorfields-private/blog-articles/looking-after-your-eyes-in-the-sun. (Accessed June 13, 2024.)
Originally published September 19, 2017. Reviewed and updated April 2022 and June 2024.
You made it to the end—nice work! If you’re the kind of scientist who likes figuring things out without wasting half a day on trial and error, you’ll love our newsletter. Get 3 quick reads a week, packed with hard-won lab wisdom. Join FREE here.
Put this article into practice
Choose a free resource to help you move forward
PROTOCOL
Chemically Competent Cells Protocol
CHEAT SHEET
Nuclear Extraction Protocol








