Phosphorylation Equals Cell Signaling!
How do cells communicate and respond to their environmental cues? This question has been on the hot list for scientists ever since the discovery of the cell. Cells use signaling cascades based on biochemical reactions to deliver or receive messages. How cool is that?
The major secret of cell signaling was discovered by a group of very talented scientists.1 They found that cells use a process called “protein phosphorylation”: the addition of a phosphate group to certain amino acids of proteins, to transduce signals from the extracellular environment. Figure 1 illustrates the complexity of cell signaling events using the tumor necrosis factor receptor signaling pathway as an example. Keep in mind that this is only one example—there are hundreds of receptors on the surface of a cell!
So, how does phosphorylation work in cell signaling? Phosphorylation is involved in the activation or inactivation of certain proteins. Out of 20 amino acids that our bodies use, three amino acids can be phosphorylated for signaling transduction pathway. Serines, Threonines, and Tyrosines all have a hydroxyl (OH-) side group that’s available for phosphorylation.
Today, I am going to tell you why you should put your old trustworthy western blot aside and give multi-parameter flow cytometry a try when it comes to cell signaling analysis.
Put this article into practice
Choose a free resource to help you move forward
DOWNLOAD
Blood Collection Tube Chart
EBOOK
Gene Editing 101

How Do You Detect Protein Phosphorylation?
There are many assays to detect protein phosphorylation, which all use monoclonal antibodies to detect the phosphorylated proteins. However, different methods have differing degrees of resolution and speed of the experiment.
For example, you can run western blot analysis to differentiate the phosphorylated and un-phosphorylated proteins. This method requires two separate blots: one blot detects the total amount of the protein and the second only for the activated, or phosphorylated, form of the protein. This requires more samples, which might be limited. You might have to co-immunoprecipitate to enrich a cell population before you do the western blot.
Another disadvantage about western blots is bulk analysis. Bulk analysis does not tell you if the specific populations of cells that are undergoing cellular signaling changes, unless you manage to obtain a 100% pure cell population. Therefore, sometimes detecting the activation of the signaling proteins is difficult. But there is a better solution. Flow cytometry is here to help!
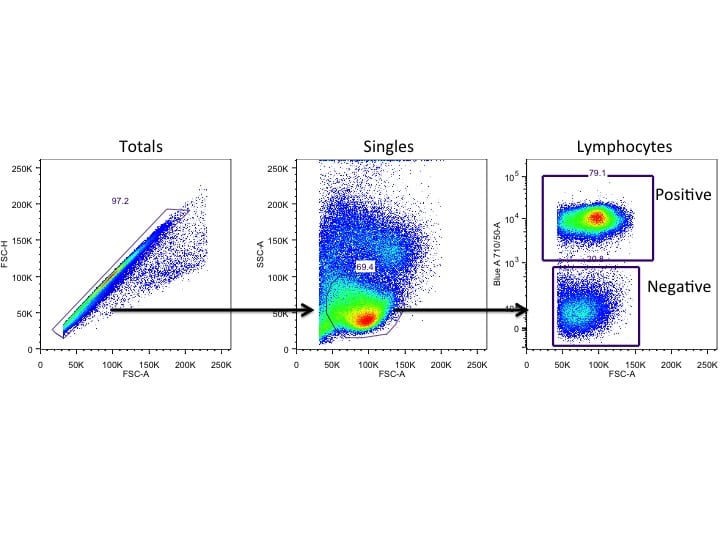
Multi-Color, Multi-Parameter Flow Cytometry for Cell Signaling
Flow cytometry, unlike other methods, provides high resolution at a single cell level. A flow cytometer records multiple parameters (depending on the capacity of the machine and the number of antibody markers available). When you are analyzing your results, you will not just find out if there is a positive band in your activated population versus a negative band in your untreated group like you would in a western blot. You can pinpoint exactly which cells are activated or if there are multiple pathways being activated.
Phosphoflow
One of the cool things with flow cytometry is that not only can you stain the surface molecule with antibodies, but you can also detect proteins inside the cell using intracellular staining protocols. In phosphoflow, peripheral blood mononuclear cells (PBMCs) or perhaps other cell populations are stimulated first with a ligand to a specific receptor. Then, the cells are fixed using paraformaldehyde to preserve the state of the proteins within the cell. Detergents permeabilize the cell membrane, which allows fluorescently labeled antibodies to enter the cell and bind to the phosphorylated proteins.
Why Is Phosphoflow Advantageous in Your Research?
Phosphoflow allows you to investigate multiple activated proteins at the same time. For example, you can examine the differences in cancerous versus healthy lung cells and run both cell types in one setting.3 Running the cells in one setting helps with efficiency and minimizing experimental variables. Also, the multiplex nature of phosphoflow means that you could look for different activated kinases using flow cytometry.5
In addition, if you use a flow cytometer that has a high-throughput system4, you can use phosphoflow in drug discovery to screen for compounds that either enhance or inhibit certain signaling pathways. So go on and give phosphoflow a try and see for yourself.
Our cells relay many different signals through receptors on the cell surface, through intermediate signaling cascades using phosphorylation as a flag (signal), and ultimately leads to activation of different genes for survival. I have provided a few pros and cons between doing traditional western blot and phosphoflow. If you are interested in detecting only a few phosphorylated proteins in the cell-signaling cascade, western blotting will definitely do the trick. However, if you are faced with a sample that contains many different cell populations, flow cytometry is a better tool. Go with the Flow!
References
- Cohen, P. (2002) The Origins of Protein Phosphorylation. Nat Cell Biol. 4(5) : E127–30. doi:10.1038/ncb0502-e127.
- Krutzik PO, Nolan GP. (2003) Intracellular Phospho-Protein Staining Techniques for Flow Cytometry: Monitoring Single Cell Signaling Events. Cytometry A. 55 (2): 61–70. doi:10.1002/cyto.a.10072.
- Irish, JM, Kotecha N, Nolan GP. (2006) Mapping Normal and Cancer Cell Signalling Networks: Towards Single-Cell Proteomics. Nat Rev Cancer. 6(2): 146–55. doi:10.1038/nrc1804.
- Chow S, Patel H, Hedley DW. (2001) Measurement of MAP Kinase Activation by Flow Cytometry Using Phospho-Specific Antibodies to MEK and ERK: Potential for Pharmacodynamic Monitoring of Signal Transduction Inhibitors. Cytometry. 46(2): 72–78.
- Perez, OD, Nolan GP. (2002) Simultaneous Measurement of Multiple Active Kinase States Using Polychromatic Flow Cytometry. Nat Biotechnol. 20(2): 155–62. doi:10.1038/nbt0202-155.
- Sanguine Bio Researcher Blog.Using Phospho-Flow Cytometry to Study Signaling in Human PBMCs. Accessed February 3, 2016.
- BD Biosciences. Phosflow™ Protocol for Human PBMCs.
- BD Biosciences. Phosflow™ Protocols for Mouse Splenocytes or Thymocytes.
You made it to the end—nice work! If you’re the kind of scientist who likes figuring things out without wasting half a day on trial and error, you’ll love our newsletter. Get 3 quick reads a week, packed with hard-won lab wisdom. Join FREE here.
Put this article into practice
Choose a free resource to help you move forward
DOWNLOAD
Blood Collection Tube Chart
DOWNLOAD
Nanodrop Results Interpretation Card