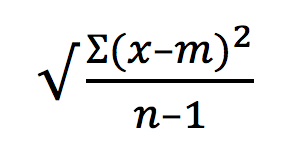Long before “Alexa” was a household name, Alexa dyes were an established series of fluorescent dyes. The inventor Richard Paul Haugland named the dyes after his son Alex. Originally a trademark of Molecular Probes, the Alexa family is now a part of Thermo Fisher Scientific.
Alexa dyes are frequently used as labels in fluorescence microscopy, flow cytometry, and other fluorescence-based applications.
Properties
The Alexa family covers a wide range of excitation and emission spectra from visible to infrared light and are classified according to their excitation maxima (Table 1).
While their chemical structures haven’t been reported yet, Alexa dyes are generally synthesized through sulfonation of dyes such as fluorescein, cyanine, coumarin, and rhodamine. This sulfonation makes Alexa dyes negatively charged and hydrophilic.
Enjoying this article? Get hard-won lab wisdom like this delivered to your inbox 3x a week.

Join over 65,000 fellow researchers saving time, reducing stress, and seeing their experiments succeed. Unsubscribe anytime.
Next issue goes out tomorrow; don’t miss it.
Alexa dyes are commercially available as amine, thiol, or sulfhydryl-reactive intermediates which can be conjugated to proteins in the lab, since proteins have primary amines and sulfhydryl groups that can be readily labeled. Oligonucleotides can also be labelled when they are synthesized with either an amine or sulfhydryl-reactive group.
Table 1: Information about features of Alexa dyes obtained from Thermo Fisher Scientific.
Name | Absorption max (nm) | Emission max (nm) | Color |
Alexa Fluor 350 | 346 | 442 | Blue |
Alexa Fluor 405 | 401 | 421 | Blue |
Alexa Fluor 430 | 433 | 541 | Green/Yellow |
Alexa Fluor 488 | 496 | 519 | Green |
Alexa Fluor 532 | 532 | 553 | Yellow |
Alexa Fluor 546 | 556 | 573 | Orange |
Alexa Fluor 555 | 555 | 565 | Orange |
Alexa Fluor 568 | 578 | 603 | Orange/Red |
Alexa Fluor 594 | 590 | 617 | Red |
Alexa Fluor 610 | 612 | 628 | Red |
Alexa Fluor 633 | 632 | 647 | Far-red |
Alexa Fluor 635 | 633 | 647 | Far-red |
Alexa Fluor 647 | 650 | 665 | Near-IR |
Alexa Fluor 660 | 663 | 690 | Near-IR |
Alexa Fluor 680 | 679 | 702 | Near-IR |
Alexa Fluor 700 | 702 | 723 | Near-IR |
Alexa Fluor 750 | 749 | 775 | Near-IR |
Alexa Fluor 790 | 784 | 814 | Near-IR |
Emission from Alexa dyes can be detected by fluorescence microscopy with appropriate filters. As you can see in Figure 1, there is an impressive range of emission wavelengths provided by this family of dyes!
Preparation of Samples Using Alexa Conjugated Secondary Antibodies
Alexa conjugated samples should be stored in the dark when not in use. This is because light exposure can result in photobleaching and poor performance of the dye. During the conjugation step, samples should be incubated at room temperature for the designated amount of time provided by the manufacturer, or at 4°C for longer durations such as overnight.
Alexa conjugated samples have excellent signal amplification and are usually diluted in blocking buffer to reduce non-specific binding. Very low concentrations of Alexa Fluor IgG conjugate can be used for fluorophore- and biotin-labeled antibodies and tandem-labeled antibodies in immunohistochemical applications.
Alexa dyes are also compatible with common fixatives such as paraformaldehyde and susceptible to typical quenchers such as trypan blue.
Applications
Alexa dyes are designed for diverse imaging applications, including:
- Live imaging and fixed cell analysis: Alexa dyes are resistant to photobleaching even when subjected to high-intensity laser sources. This makes Alexa dyes particularly suitable for cell microscopy.
- Fluorescence resonance energy transfer (FRET): The broad spectra of Alexa dyes provides several options for FRET when conjugated to proteins or oligonucleotides. Overlapping emission spectra are also useful for sophisticated confocal microscopy and when investigating multiple labels using pseudocolor techniques.
- Immunohistochemical investigations: Alexa Fluor-conjugated secondary antibodies are ideal for multiplexing because of their high specificity and sensitivity.
- Immunofluorescence: Certain Alexa dyes emit red fluorescence and are therefore the best choice for immunofluorescence in deep-red spectra.
Advantages
Alexa dyes represent a major development in fluorescence applications since they have many advantageous qualities, including:
- Brightness: Alexa Fluor conjugates exhibit more brightness than other spectrally similar conjugates. Thus, they require less total sample in experiments while enabling detection of low abundance targets.
- Photostability: Alexa Fluor conjugates possess more photostability than other conjugates, allowing more time to capture images.
- Chemical stability: Alexa dyes remain highly fluorescent over a wide pH range of 4 to 10.
- Solubility in water: Conjugation to targets can be done without using organic solvents, and the resulting conjugates are resistant to precipitation during storage.
- Compatibility: Alexa dyes can be used with corresponding standard dye filters, excitation sources, and instruments.
- Emission diversity: Alexa dyes are readily available with a wide range of fluorescent colors from blue to red.
Alternatives to Standard Dyes
Alexa dyes can replace many standard dyes after careful consideration. If you are looking to switch to Alexa dyes, you should compare characteristics including absorption and emission spectra, Stokes shift, pH-sensitivity, stability to photobleaching, extinction coefficient, and quantum yields.
Importantly, all Alexa dyes are sulfonated forms of different basic fluorescent substances, so they are often similar to standard dyes in their excitation and emission maxima (Table 2).
Table 2: Information about Alexa dyes as alternatives to the common dyes obtained from Thermo Fisher Scientific.
Common dyes | Alexa dyes |
AMCA (coumarin) | Alexa Fluor 350 |
Cy2, FITC (fluorescein) | Alexa Fluor 488 |
Cy3, TRITC (rhodamine) | Alexa Fluor 555 |
Rhodamine red | Alexa Fluor 568 |
Texas Red | Alexa Fluor 594 |
Cy5 | Alexa Fluor 647 |
Cy5.5, IR680 | Alexa Fluor 680 |
Cy7 | Alexa Fluor 750 |
With that said, Alexa dyes are often more expensive than common dyes. Series of fluorescent dyes similar to Alexa Fluor such as DyLight Fluor and Atto dyes are also available.
While it can be challenging to select the best dye for a particular application, having a wide range of dyes to choose from is very exciting (no pun intended!). Always perform empirical testing to validate new dyes, whether they be from the Alexa family or another!
References:
- Gregory, Jay, ed. Handbook of Fluorescent Probes and Research Products, 1st Eugene, OR. Molecular Probes Inc. 2002.
- The Alexa Fluor Dye Series Technical Notes, Thermo Fisher Scientific.
You made it to the end—nice work! If you’re the kind of scientist who likes figuring things out without wasting half a day on trial and error, you’ll love our newsletter. Get 3 quick reads a week, packed with hard-won lab wisdom. Join FREE here.