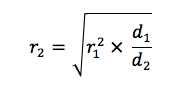Bacterial cultures may be much easier to grow than mammalian cells, but if your yields are suboptimal there are plenty of parameters to play with. Here we list a few of the things you should consider to maximize your culture growth.
Shaking speed
Shaking is performed to allow aeration of your culture, which is of course important to get high yields. But what is the optimum speed to shake at? This is something that you need to play around with and determine for your own experiments. In general, higher revolutions per minute (RPM) will increase the amount of oxygen, but be careful not to ramp up the speed too high, as this could cause damage to your cells either physically or by potentially generating oxidative stress1. Often a speed of around 200–250 RPM is used.
Not all shakers are equal
If you’ve perfected your shaking speed be aware that if you move to a new shaker (for example if you move labs or your replace your old shaker) you’ll have to compensate for differences in the orbital diameter of the shaker to achieve the same oxygen transfer rate. Luckily there is a simple equation to help you2:
Put this article into practice
Choose a free resource to help you move forward
CHEAT SHEET
SDS-PAGE Protocol Cheat Sheet
EBOOK
Free Guide to Protein Expression
Where r1 = the rpm for the old shaker, r2 = the rpm for the new shaker, d1 = the orbital diameter for the old shaker and d2 = the orbital diameter for the new shaker.
Use a Baffled Flask
A baffled Flask. Source: Kjaergaard.
No these flasks aren’t confused! Instead they are flasks that include ‘baffles’ that work to interrupt the laminar flow of your media. How does this help? Well by creating turbulence you increase the amount of oxygen take-up into the media and more oxygen means more growth!
Don’t have a baffled flask? Get your shake on!
Not having baffled flask isn’t the end of the world; you just need to shake your flasks a little faster to ensure good aeration.
Cover It up Carefully
Back in my lab we would cover our flasks with aluminium foil, while other labs would use cotton plugs. However, what you use to cover your flasks will have an impact on how much oxygen they get. Not covering your flasks at all will provide the most aeration, but this is impractical as it leaves the door wide open for cross contamination of samples. Instead avoid tightly sealing your flasks with cotton or consider using a breathable membrane, which provides protection from contamination but is permeable to oxygen and carbon dioxide.
Turn Down the Volume
One thing that baffled me when I first saw bacterial cultures is why do people use such large flask but hardly any media? Despite my initial thoughts that people were being wasteful or there was a shortage of smaller flasks, the truth is that your flask volume should be at minimum five times the volume of the amount of media you are using. There are two main reasons for this:
- The first is, unsurprisingly, oxygen – too much volume will limit the amount of oxygen that your cultures get.
- The second is to prevent spilling of cultures, which can cause cross contamination.
Add Some Magnesium
Magnesium is required for many bacteria to grow3 and therefore addition of magnesium either as MgCl2 or MgSO4 can improve the growth of your cultures, especially if you use basic media such as LB broth. Some other media, such as SOB and SOC already contain magnesium, and therefore addition of magnesium is not required. You can find recipes for different culture media in our article Pimp Your Plasmid Growth Medium.
What do you do to maximize your culture growth?
Further reading:
1. Munna MS, Tamanna S, Afrin MR, Sharif GA, Mazumder C, Kana KS, et al. (2014) Influence of Aeration Speed on Bacterial Colony Forming Unit (CFU) Formation Capacity. Am J Microbiol Res. 2:47–51.
2. Bates MK, Phillips D, O’Bryan J. Predicting Optimal Bacterial Growth. Laboratory Equipment.
3. Webb M. (1949) The Influence of Magnesium on Cell Division The Effect of Magnesium on the Growth of Bacteria in Simple Chemically Defined Media. J Gen Microbiol. 3:418–24.
4. Klöckner W, Büchs J. (2012) Advances in shaking technologies. Trends Biotechnol. 30:307–14.
You made it to the end—nice work! If you’re the kind of scientist who likes figuring things out without wasting half a day on trial and error, you’ll love our newsletter. Get 3 quick reads a week, packed with hard-won lab wisdom. Join FREE here.
Put this article into practice
Choose a free resource to help you move forward
EBOOK
Free Guide to Protein Expression
CHEAT SHEET
SDS-PAGE Protocol Cheat Sheet