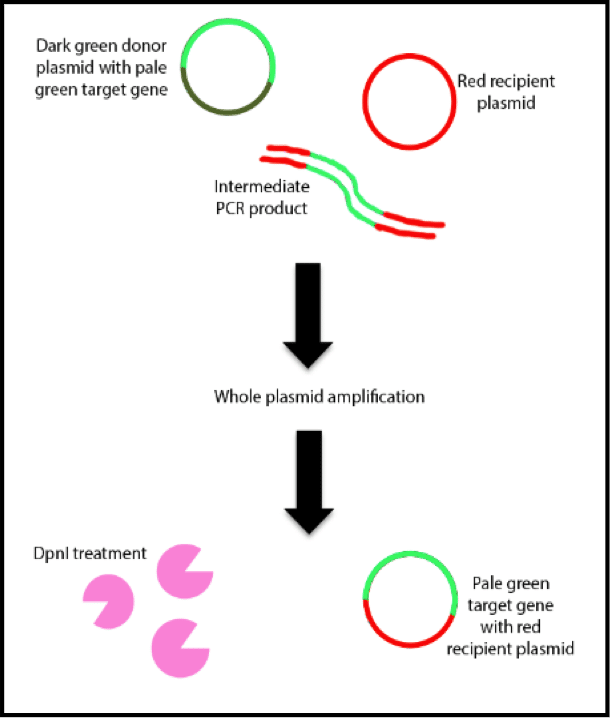
If you’re picking up a list of available plasmid vectors for the first time, it can be mind-blowing to decide on the best one for your experiment. What cloning sites do you need? What restriction enzyme sites? What insert size is possible?
Let me help you with some pointers about what factors to look out for.
Since the appearance of the first commercial cloning vectors in the 70s, the number of products available has increased at an astounding rate. However, despite this incredible variety, the secret that you need to know is that there are only a few parameters relevant to choose the best plasmid.
Insert Size: large or small?
The only aspect to consider here is whether you’re cloning a large or small DNA fragment. Most general plasmids (i.e. pUC and pBluescript) can cope with inserts up to 15 kb. Anything bigger can complicate replication and cause problems with stability. Don’t despair if you have a large DNA insert, however, as there are special types of vectors suitable for such applications.
Enjoying this article? Get hard-won lab wisdom like this delivered to your inbox 3x a week.

Join over 65,000 fellow researchers saving time, reducing stress, and seeing their experiments succeed. Unsubscribe anytime.
Next issue goes out tomorrow; don’t miss it.
Copy Number: high or low?
In most cases, a high-copy plasmid vector is the best approach to produce the highest yields. For example, pBluescript has a copy number 300-500 and pUC can reach 700. You may be wondering, then, why some plasmids have a more modest value of 10-12 copy number, as is the case with pACYC. These more specialised vectors have been developed to counteract some problems caused by high-copy plasmids. Your DNA fragment may, for example, become toxic to the cell when present in high levels. In this case, the best way to avoid any problems may be to use a low-copy vector.
Cloning Sites: which restriction enzymes?
A crucial step during the cloning process is the ligation of the DNA fragment to the plasmid. This is greatly facilitated by the use of specific restriction endonucleases. This means it’s important to check whether plasmid and selected restriction enzymes are compatible. In reality, this is not so problematic nowadays, as most modern vectors include an artificial stretch of DNA with many different restriction endonuclease cutting sites. If all else fails, blunt-end ligations are possible, but can be very difficult to complete successfully.
Check out our related article to learn how to read plasmid maps, including how to find the restriction sites.
Antibiotic resistance: why is it needed?
It shouldn’t come as a surprise that introducing foreign DNA (i.e. your carefully prepared plasmid) into cells is an inefficient process. To add insult to injury, cells that have not incorporated the plasmid are at an advantage, as they do not have to replicate additional DNA (this may be particularly relevant when dealing with a large DNA fragment).
This means that, without the ability to discriminate which cells are positive, you may be looking at a dish full of healthy colonies … but none with the plasmid. This is where antibiotic resistance comes in. Almost all conventional plasmids are designed in such a way that only the cells that have incorporated the vector can survive in the presence of a specific antibiotic. All you have to do is chose which antibiotic to use – ampicillin, tetracycline or kanamycin.
A few criteria to avoid a headache!
So, it may seem daunting to choose the right plasmid, but following these simple criteria can quickly guide you to select the best option for your project. Rest assured that general plasmids are suitable for most applications and most likely, you’ll only ever need a few.
You made it to the end—nice work! If you’re the kind of scientist who likes figuring things out without wasting half a day on trial and error, you’ll love our newsletter. Get 3 quick reads a week, packed with hard-won lab wisdom. Join FREE here.