Ever thought about using a kit-free DNA gel extraction method?
Kits for DNA gel extraction are a great way to save time in the lab, but they are costly and can produce a lot of plastic waste. If you are a PhD student, maybe even more important is that using kits stops you from thinking about what they are doing and WHY.
You can start saving money and reducing waste by recycling your DNA extraction columns. But this would be a half-measure. Going kit-free can save you money, reduce waste, get your brain cells active, and help you troubleshoot when necessary or improve the protocols.
How DNA Extraction Kits Work
In the case of DNA fragment isolation, there’s an initial stage of separating the fragment(s) of interest from the rest of your DNA by running the DNA on a gel that separates them by size. Then, after visualizing the fragments and comparing them to a size standard, you cut the band(s) of interest, dissolve the agarose and proceed with the extraction.
In a nutshell, the DNA extraction kits work in three steps: bind, wash, and elute. DNA should be extracted from the agarose gel, bound to a substrate, and eluted. Depending on the elution protocol, precipitation and resuspension in the buffer suitable for downstream applications may be needed. Note that PCR usually tolerates less purified samples than cloning, e.g., you don’t need to reprecipitate your DNA after gel extraction if you use it as a template.
A consideration for all methods is to minimize the size of the fragment. A larger fragment means more contamination and more dilute DNA as a result.
Three Easy Kit-free DNA Gel Extraction Methods
Method 1: Bind to Paper
In this method, 3MM filter paper serves like a nitrocellulose membrane in Southern or northern blot binding DNA, except the transfer via an electrical field is not vertical but horizontal. (1)
In this kit-free method, you cut the gel upstream of the running direction and insert a piece of filter paper, after visualizing your DNA in the gel. Continuing the electrophoresis binds the fragment to the paper. You’ll then need to wash your bound-to-paper DNA fragment and elute it using a tris-EDTA (TE) buffer or water.
Washing is done by placing the paper fragment in a 0.5 ml tube and adding 50–100 μl of TE.
Next, carefully make a hole in the bottom using a syringe needle, place it in a 1.5 ml tube, and spin. The buffer with eluted DNA collects at the bottom of the bigger tube.
Repeating the elution increases the amount of DNA you recover and its volume, so you may need to ethanol precipitate your DNA.
The advantage of this method is that it doesn’t require much, except for some filter paper and tubes. But the time it takes to bind DNA depends on the size of the fragment, gel density, and voltage. You need to standardize conditions and may discover that your fragment has run away from you, and you need to repeat the procedure.
But you can avoid the guesswork of transfer time by using one of the methods below, which doesn’t require casting a net in the hope of catching your DNA like a fish.
Method 2: Using Glass Wool or Cotton Wool
This method requires two centrifuge tubes—one 0.5 ml and one 1.5 ml—plus some glass wool or cotton wool (just plain bleached cotton you find on cotton wool buds). (2)
Punch an inside-to-out hole in the smaller tube and insert it into the bigger tube.
Put some of your chosen wool on the bottom of the tube.
Place the fragment on the wool cushion and close the lid of the smaller tube.
Spin the tube in a microcentrifuge for 5–10 minutes, depending on the size of the fragment:
- 1 kb: 5,000 rpm for 5 min
- 1–2 kb: 10,000 rpm for 5 min
- 2–3 kb: 10,000 rpm for 10 min.
The buffer with dissolved DNA filters through the wool or cotton during the spin and collects in the outer tube. Depending on the size of the initial gel fragment, the recovery volume is 15–30 μl.
The authors of the method did not experiment with fragments larger than 3 kb. But considering that the recovery rate falls as fragment size increases, it is probably not worth doing.
Method 3: Using Dialysis
Dialysis is another simple way to get your DNA back from a gel. (1)
First, freeze the fragment at –20°C for 30 min.
Meanwhile, soak a dry piece of dialysis tubing in distilled water, place it into the same buffer as your fragment (e.g., TBE), and seal one end.
Place the fragment into the tubing. Add 200–400 μl of TBE and seal the other end.
Place into the electrophoresis tank and apply 5V/cm current. The DNA comes out of the gel fragment but stays inside the tubing. After 10–15 minutes, unseal one end of the tubing and remove the buffer inside that contains your DNA. It then can be precipitated.
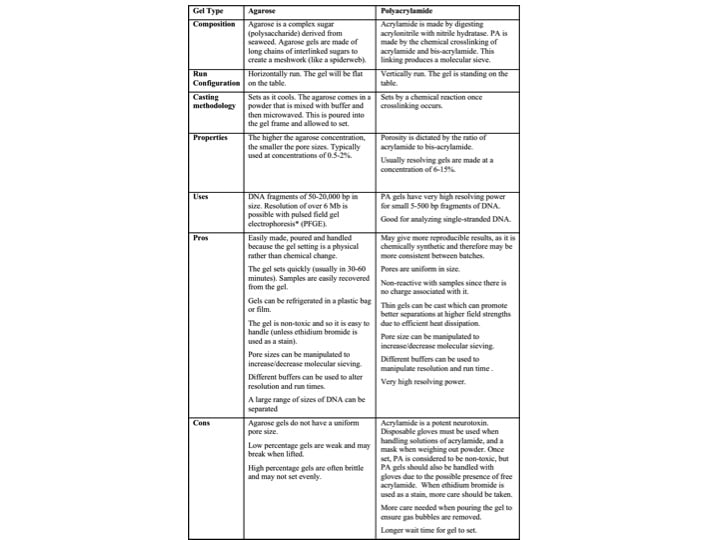
DNA Gel Extraction Methods Summarized
Table 1 compares the cost, difficulty, and recovery for different DNA gel extraction methods.
Table 1. Comparison of methods for kit-free DNA gel extraction
Method | Cost (per sample) | Difficulty | DNA recovery | Downstream ethanol |
Spin columns | +++ | + | 70–90% | – |
Paper | + | ++ | 20–90% | + |
Cotton wool | + | + | 30–60% | – |
Glass wool | ++ | + | 30–60% | – |
Dialysis | ++ | +++ | 70–90% | + |
For more help with your DNA gel extractions, read our related article and get 10 DNA gel extraction tips to maximize your extractions.
For more tips, tricks, and hacks for getting your experiments done, check out the Bitesize Bio DIY in the Lab Hub.
Do you use any of the above DNA gel extraction methods, or perhaps reclaim your gel-bound DNA another way? Let us know in the comments below!
References
- Lewis, M. DNA extraction from agarose gels (basic method). MethodBook Accessed 01/06/2022.
- Sun, Y, et al. (2012) A quick, cost-free method of purification of DNA fragments from agarose gel. Journal of Cancer 3:93–95.
You made it to the end—nice work! If you’re the kind of scientist who likes figuring things out without wasting half a day on trial and error, you’ll love our newsletter. Get 3 quick reads a week, packed with hard-won lab wisdom. Join FREE here.
Put this article into practice
Choose a free resource to help you move forward
CHEAT SHEET
Guide to Pipette Cleaning
EBOOK
Guide to Lab Safety