Hello again, fellow Flow Cytometry Fan! It looks like you have your experiment all planned out, including staining protocols and gating schemes, and are ready to get some paradigm-shifting data. But before we start “plugging-and-chugging” samples through your cytometer of choice, we need to make sure that the nozzle size and sheath pressure are set up correctly.
Seriously? When Am I Ever Going to Get My Data?!
I know, I know. Running a flow cytometry experiment is super fun and exciting, but preparation can be tedious. Annoying as it is, I swear that all this work is worth it and absolutely necessary to ensure your data is reliable the first time around.
Let’s Talk About…Fluidics
Whether you are performing a cell sorting experiment, or just immunophenotyping, it is important that you take a peek at the nozzle size and sheath/sample pressure that you will use during your run. For most machines, the general set-up works splendidly for most samples. However, you’re a Super Scientist and know that it’s better to know than not, especially if it turns out that you need a different set-up because your samples are a bit finicky. Knowledge is power!
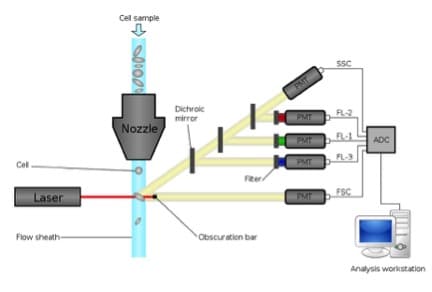
As a recap, a cytometer has 3 main components: fluidics, optics, and electronics systems. While the cooperation and function of all three are imperative to get out any data at all, the fluidics system is probably the most overlooked in terms of importance. But like with any experiment, we have to be sure to start off on the right foot! The whole purpose of the fluidics system is to get the sample into the machine so that it can be interrogated by laser beams. And to make sure that this goes off without a hitch, we have to be sure we’re using the correct nozzle size and sheath/sample pressure.
Put this article into practice
Choose a free resource to help you move forward
DOWNLOAD
Blood Collection Tube Chart
CHEAT SHEET
Lab Math Cheat Sheet
Nozzles for Daaayzzz
Most may not realize it, but every flow cytometer comes with at least 3 – 4 nozzles! This allows users to investigate a wide range of particle/cell sizes. Nozzles are typically available in 70, 85, 100, and 130 µm. Most lymphocytes and suspension cells are run using a 70 µm nozzle, while larger cells, including adherent cell lines and tissue dissociations, and more delicate cells are investigated with a 100 µm nozzle (Table 1). The general rule of thumb is that your nozzle size should be about 4-5 times larger than the size of the cells being interrogated.
Nozzle Size | Cell Type Example | Sheath Pressure |
70μm | splenocytes, non-activated T cells, B cells, platelets, bacteria, yeast | High (70 psi) |
85μm | cell lines, activated T cells, plasma cells, NK cells | Medium (45 psi) |
100μm | DCs, macrophage, granulocytes | Low (20 psi) |
So Much Sheath Pressure!
Once your sample enters the cytometer via the nozzle, it is injected into a stream of sheath fluid (essentially saline solution) within an area called the “flow chamber”. The flow chamber is designed to, in an ideal situation, place the sample particles in the center of the sheath fluid stream (where the laser beam is focused) and only one particle should move through the laser beam at a time (here is a really helpful diagram).
Regardless of which type of cytometer you’re using, there are 2 types of pressures to be aware of: sample pressure and sheath fluid pressure. Both are needed because the success of getting the sample through the machine depends on principles relating to laminar flow – meaning that the sample core (and thus sample pressure) remains separate but coaxial within the sheath fluid. The flow of sheath fluid accelerates the particles and restricts them to the center of the sample core. This process is known as hydrodynamic focusing.
Sample Pressure
If this all seems a bit new, have no fear! You’re likely more familiar with sample pressure. The sample pressure regulator controls the sample flow rate by changing the sample pressure relative to sheath pressure. If you’ve ever worked on a BD benchtop cytometer (LSR, Fortessa, etc.) and have noticed those buttons labeled “LO”, “MED”, and “HI”? Those are fixed sample pressure settings for ease of use. You may also have noticed that when you select “HI”, a lot more cells per minute are analyzed, and thus you’re able to run your sample set much faster.
So why not just run your samples on “HI” all the time? Well, increasing the sample pressure increases the flow rate by increasing the width of the sample core (aka the sheath fluid stream carrying your cells through the machine). This, in turn, allows more particles to enter the stream within a given moment. With a wider sample core, some particles could pass through the laser beam off-center and intercept the laser beam at a less optimal angle. For some, this isn’t that big of a deal. For instance, many times immunophenotyping experiments are run at a higher flow rate because the user is not as concerned with resolution. However, if you are sorting cells that are going to be analyzed for DNA, it is in your best interest to run at a lower flow rate to make sure that the majority of the cells pass through the center of the laser beam.
As the saying goes, the devil is in the details, and this is no truer than when dealing with flow cytometry. And now that you are armed with more knowledge than you know what to do with, you can be over the moon confident in your experiment. Now go forth and capture that mind-blowing data!
You made it to the end—nice work! If you’re the kind of scientist who likes figuring things out without wasting half a day on trial and error, you’ll love our newsletter. Get 3 quick reads a week, packed with hard-won lab wisdom. Join FREE here.
Put this article into practice
Choose a free resource to help you move forward
DOWNLOAD
Blood Collection Tube Chart
EBOOK
The Fundamentals of qPCR and RT-qPCR