No, this is not a call for Geeks to take over the world – just a tiny part of it – the part that ensures success in all experiments or at least a good way to analyze them if they fail.
There are all too many entry points for error and variability that can be introduced into experimental measurements – and it’s critical to control those points. This is especially true in Flow Cytometry where there are multiple controls essential for proper interpretation of the data.
Flow cytometry controls can be divided into 5 major classes. The purpose of these controls to ensure the instrument is performing properly, that compensation is correctly set, that appropriate gates can be drawn and the correct interpretation of your data is possible.
Instrument controls
These controls are used to monitor the performance of the flow cytometer. The operator of the instrument often runs this class of controls on a regular basis. Instrument controls check the following:
Put this article into practice
Choose a free resource to help you move forward
DOWNLOAD
Blood Collection Tube Chart
POSTER
Histological Stains Poster
- PMT sensitivity
- Fluidics
- Laser Delay
These controls are imperative to ensure that the instrument is performing correctly and that any changes found in the samples are not due to instrumentation issues.
Compensation controls
Compensation controls are used to set proper compensation. The compensation controls, be they beads or cells, should meet and follow the three rules of compensation:
- Controls need to be at least as bright as any sample they will be applied to
- Background fluorescence should be the same for the positive and negative control populations for any given parameter
- Experimental sample and control samples must be matched
- Identical fluorochrome
- Identical instrument sensitivity
Proper compensation is key for proper interpretation of the data.
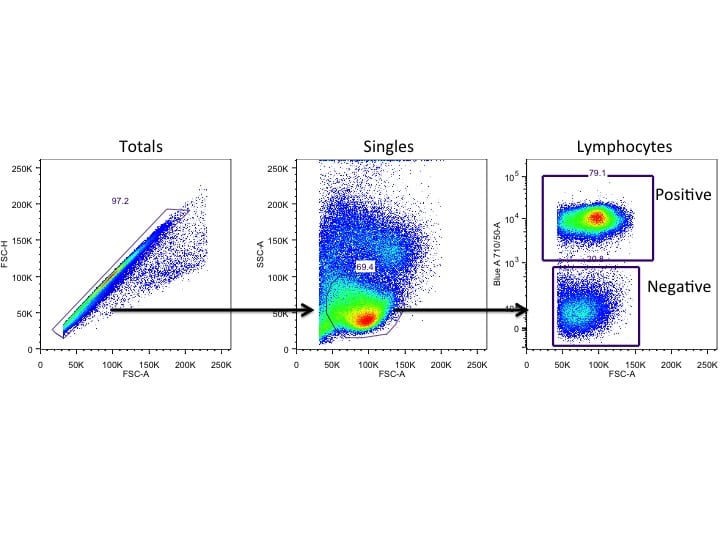
Gating controls
Proper gating controls are used to identify the populations of interest. The most common of these controls is the Fluorescence Minus One (FMO) control. This is used to ensure proper placement of gates for analysis.
Non-specific binding controls
Non-specific binding (NSB) reduces the sensitivity of measurement in flow cytometers. There are no perfect controls to measure NSB, but the isoclonic control is a good way to ensure that the antibody being used is actually binding to the target. Specific binding can be outcompeted, while NSB cannot.
Some researchers still include the isotype control in this control class. The idea of the isotype control is that an antibody with the same isotype but a different target can be used to determine NSB. Unfortunately, this is not the case, and for the most part, the isotype control is not a useful control.
Experimental controls
The last major class of controls refers to anything that is used to determine if the treatments or conditions have caused a true change. These controls include untreated or unstimulated controls to identify the background levels of expression, stimulated controls to ensure stimulation conditions worked and reference controls to monitor the process during long-term studies.
Are all these controls really needed?
The short answer is – YES.
The longer answer is that during the developmental phases of a cytometry panel, it is critical to use these controls to identify the sources of error that are necessary to have controls for. For example, most groups will not run FMO controls for well-characterized populations like viability dyes or CD3. However, a well controlled experiment uses all these controls to ensure a proper interpretation of the data.
For those wanting more information, stay tuned to this channel for more detail on these control classes. Finally, check out the following reference for thought – Hulspas et al. for more information on controls.
References
- Hulspas et al. (2009) Considerations for the control of background fluorescence in clinical flow cytometry. Cytometry B. 76B:355-364.
You made it to the end—nice work! If you’re the kind of scientist who likes figuring things out without wasting half a day on trial and error, you’ll love our newsletter. Get 3 quick reads a week, packed with hard-won lab wisdom. Join FREE here.
Put this article into practice
Choose a free resource to help you move forward
DOWNLOAD
Blood Collection Tube Chart
POSTER
Antibiotics Reference Guide