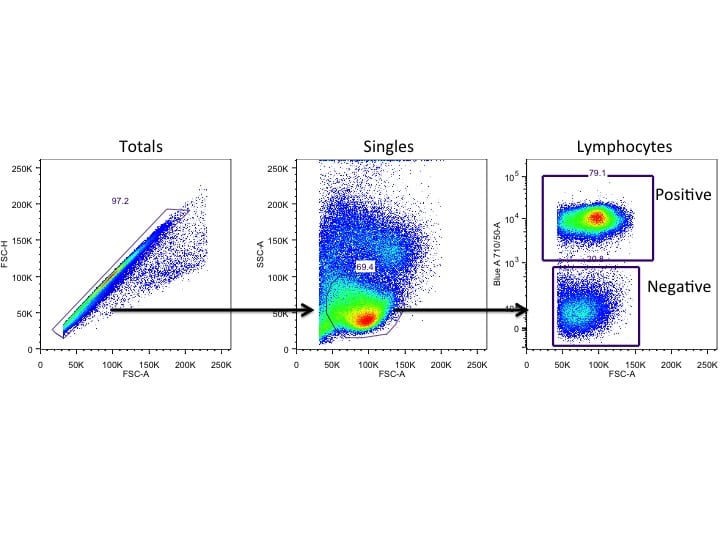
One of the key characteristics of cytotoxic cells (i.e. CD8+ T cells, natural killer cells) is the presence of pre-formed cytoplasmic lysosomal granules. These structures house perforin and granzyme; two molecules that are essential for the lysis of target cells. Upon effector cell activation, granules are polarized toward the target cell and the contents are deposited, likely through the immunological synapse, resulting in target cell lysis. Thus, in studying immunological responses, measuring cellular degranulation can be one of the most telling pieces of information regarding how cytotoxic effector cells are functioning. However, this can seem a daunting task since the granules are recycled incredibly quickly – so how is one to ever determine the ability of their cell of interest to degranulate? Good question! It’s actually quite easy. The key is to be quick yourself – with staining, that is.
As the contents of the granules are deposited toward the target cell, the proteins that form the internal lipid bilayer of the granule are exposed to the extracellular space. These proteins are called lysosomal-associated membrane proteins (LAMPs), with one of the most common being LAMP-1, or CD107a. Thus, a successful degranulation assay measures the degranulation process of the cytotoxic cell by detecting CD107a expression via flow cytometry.
Catching CD107a
Unfortunately, the extracellular expression of CD107a is extremely transient owing to the recycling of the granules, so you must stain for CD107a during stimulation of your effector cells before you have performed your surface stain. Another trick with a degranulation assay is being specific with the detection of CD107a. The whole idea behind this assay is to “catch” the expression of CD107a as it transiently appears during degranulation. However, if you double-stain for CD107a (i.e., adding your anti-CD107a antibody during stimulation as well as during surface staining), you run the risk of detecting CD107a that hasn’t been re-internalized. Furthermore, you can actually get over-detection of CD107a if you happen to add your anti-CD107a antibody to your samples after they have been permeabilized, as in the case of a simultaneous intracellular cytokine staining assay (ICS) where you may be also looking for IFNg, granzyme B and/or perforin expression in these effector cells.
Here’s your über simple, easy-as-pie protocol (along with some insider tips):
Put this article into practice
Choose a free resource to help you move forward
DOWNLOAD
Blood Collection Tube Chart
EBOOK
Curious Tales from the History of Biology
- At the same time as the stimulus is added to your cells of interest, also add your fluorescently conjugated antibody directed against CD107a and allow for your culture to incubate for 1 hour at 37°C
- Be sure that the anti-CD107a antibody you use is directly conjugated to a fluorophore. Trying to deal with secondary staining of such a transiently expressed protein is going to give you an unnecessary headache.
- Don’t forget a positive control! This is going to be the crux of interpreting your data later on and will depend on the effector cell type you’re investigating. For instance, while PMA/ionomycin works just fine to stimulate T cell degranulation, using an actual target cell line (K562) is the way to go for NK cells.
- Culture for another 5 hours in the presence of monensin or brefeldin A.
- Monensin and brefeldin A are two commonly used chemicals to retain cytokines in the cytoplasm for detection during an ICS. The purpose of these chemicals during a degranulation assay is to help neutralize the acidification of the granules, which would otherwise lead to quenched fluorescence.
- Monensin, which interacts with Golgi transmembrane transport, is the preferred choice when staining for only CD107a and not intracellular antibodies.
- Brefeldin A, which redistributes proteins from the Golgi to the ER, is commonly used when wanting to detect both CD107a and IFN?
- BD has a great table to help with your decision.
- It is extremely important not to allow your assay to culture for longer than 6–9 hours, since prevention of protein secretion in any fashion can result in cell death.
- Proceed with standard staining of samples.
- Some persons may choose to stain only for CD107a expression, but that would be such a waste! You should definitely be looking at coinciding expression of perforin and granzyme. Afterall, what’s the point of degranulation if there’s nothing inside the granules?
- For the love of [fill in your deity of choice here], do not under any circumstances stain for CD107a again!
Don’t forget about analysis!
Analysis of your degranulation assay should be pretty straightforward; just keep in mind what is happening biologically. In the presence of a stimulus, be it protein or target cell, your healthy effector cell of interest will degranulate, leading to (1) increased expression of CD107a (that you were able to successfully “catch” with the protocol above – go you!), and (2) a decrease in perforin and granzyme. Why the decrease? Remember: while monensin/brefeldin A prohibit secretion of newly made proteins, they have no impact on the already pre-formed granules hanging out in the effector cell cytoplasm.
One last thought
A great partnership with a degranulation assay is a chromium release assay, which will allow for analysis of actual target cell killing by your effector cell of interest. Stay tuned for the next installment!
And there you have it: your very own guide to unraveling a degranulation assay. Questions? Concerns? Tidbits of your own or things you think I missed? Comment below!
References
- Betts MR et al. (2003) Sensitive and viable identification of antigen-specific CD8+ T cells by a flow cytometric assay for degranulation. Immunol. Methods 281(1–2):65–78.
- Chan KS et al. (2007) Flow cytometric detection of degranulation reveals phenotypic heterogeneity of degranulating CMV-specific CD8+ T lymphocytes in rhesus macaques. Immunol. Methods 325(1–2):20–34.
- BD Biosciences Intracellular Flow: Cytokine Detection
Featured image from University of Michigan School of Natural Resources & Environment.
You made it to the end—nice work! If you’re the kind of scientist who likes figuring things out without wasting half a day on trial and error, you’ll love our newsletter. Get 3 quick reads a week, packed with hard-won lab wisdom. Join FREE here.
Put this article into practice
Choose a free resource to help you move forward
DOWNLOAD
Blood Collection Tube Chart
DOWNLOAD
The Happy Scientist Reference Pack