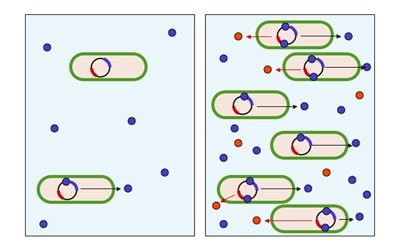
Cell Penetrating Peptides (CPPs) are the Trojan Horse of cell biology – an innocuous peptide sequence with the special ability to carry virtually any cargo across the plasma membrane. If you have a special delivery that you don’t want to get lost in transit, CPPs are for you!
CPPs are short peptides (typically 5-30 amino acids) whose main claim to fame is that they can translocate through the plasma membrane. This is unusual, as most peptides rely on active transport for their entry into the cell.
Currently the exact mechanism of how this CPP translocation is not well-defined, especially as CPPs come in a variety of shapes, sizes, charges, and sequences. Although a lot of effort has gone into defining the minimum sequence required for cellular uptake, so far all that is really known is that most CPPs have a net positive charge and/or are amphipathic (i.e. containing both hydrophilic and hydrophobic sections, much like the phospholipids that make up the plasma membrane). These properties allow CPPs to physically interact with the plasma membrane and facilitate uptake, commonly (but not always) via endocytic pathways.
Cargo Carriers
If you’re thinking about using CPPs in your research, it’s probably because you want to get something inside a mammalian cell. CPPs have been used as transporters of many different cargos, such as:
Put this article into practice
Choose a free resource to help you move forward
CHEAT SHEET
Western Blot Cheat Sheet
EBOOK
Free Guide to Protein Expression
- Nucleic acids – including oligos, siRNA, plasmids, dsDNA
- Peptides – including antibodies, substrates or inhibitors of intracellular enzymes, ligands for different protein binding domains, activators or inhibitors of transcription factors
- Contrast agents – including fluorescently-labeled peptides, and gold nanoparticles
- Drugs – including chemotherapeutics such as doxorubicin, methotrexate or paclitaxel
Generally these cargos are bound to the CPP either by expression as a fusion protein, covalent conjugation (e.g. peptide linkers or disulfide bridges) or simply by mixing the components together to form complexes. This step can be rate limiting and may require some optimization. For example, although mixing is the easiest method, each CPP has ideal mixing ratios with different cargo types to ensure efficient complex formation. Also, some cargos only form complexes with specific types of CPPs.
Since their initial discovery in the 1980s, many different CPPs have been discovered or synthesized. The two best-known CPPs are Tat and Penetratin. Lots of CPPs have been derived from these two naturally occurring peptides, so let’s take a look at these.
Tat
Derived from the HIV-1 regulatory protein, tat, that was found in 1988 to be taken up by cells in tissue culture and remain functional after uptake. The peptide contains a cationic stretch of eight arginines, which was subsequently found to be the minimal sequence for translocation of the membrane. Tat and the other polyarginine CPPs are all nonamphipathic peptides, and rely solely on the high content of positively charged amino acids for their uptake. They need to be used in higher concentrations because they don’t readily interact with the amphipathic phospholipid bilayer. These CPPs are mainly translocated via endocytic mechanisms.
Penetratin
Discovered in 1994, Penetratin is derived from a 60-mer peptide encoded by the Drosophila antennapedia gene homeobox. Penetratin refers to the 16 amino acid peptide constructed from the sequence of the third helix. This short peptide is strongly charged, like Tat and its derivatives, and is nonamphipathic. The 6 arginine and 3 tryptophan (R6W3) CPP was derived from the Penetratin minimal sequence. Like Tat, these CPPs rely on endocytic mechanisms for their uptake.
Although both of these initial CPPs were cationic, a greater understanding of the varied mechanisms of membrane translocation have allowed the use of new CPPs that are amphipathic or hydrophobic. These CPPs use different translocation methods. They are able to directly interact with the plasma membrane and use direct insertion or pore formation for their translocation into the cell.
Although you can buy single CPPs if you have a specific use in mind, most commercially available CPP products are actually CPP cocktails. This is advantageous for those who don’t know how to choose the best CPP for their target compound, as it allows a more “one-size fits all” approach. You can use the same CPP mixture with many different cargo structures. A cocktail triggers different cellular uptake mechanisms to maximise efficiency of translocation, and using CPPs mixtures maximises compatibility with different cell types.
How have you used CPPs in your research? Do you have any tips and tricks?
You made it to the end—nice work! If you’re the kind of scientist who likes figuring things out without wasting half a day on trial and error, you’ll love our newsletter. Get 3 quick reads a week, packed with hard-won lab wisdom. Join FREE here.
Put this article into practice
Choose a free resource to help you move forward
CHEAT SHEET
Western Blot Cheat Sheet
CHEAT SHEET
SDS-PAGE Protocol Cheat Sheet