“The greater the power, the more dangerous the abuse.” – Edmund Burke
“With great power comes great responsibility.” – Uncle Ben (quoting Voltaire)
While it’s unlikely that either of these speakers performed multi-dimensional flow cytometry, it is important to remember these quotes in the context of developing and implementing a good polychromatic flow panel. More fluorochromes are always desired – because that helps reveal more cellular subsets, and allows us to ask more complicated biological questions.
Optimized Multicolor Immunofluorescent Panels
The process of designing a panel can be daunting and time-consuming. For this reason, Cytometry A has recently started publishing a novel article type called “Optimized Multicolor Immunofluorescent Panel” or affectionately known as the OMIP. These articles provide details about an individual polychromatic flow cytometry panel, including what question(s) this panel was designed to answer, what reagents were used and a sample of the gating strategy. There are over 20 OMIPs published as of this writing, with a majority of them targeting T-cell phenotyping.
Put this article into practice
Choose a free resource to help you move forward
DOWNLOAD
Blood Collection Tube Chart
CHEAT SHEET
Lab Math Cheat Sheet
Designing Your Own Polychromatic Flow Panel
If there is not an OMIP that you can use to answer your research question, the hard work begins.
Step 1: Figuring out Your Experimental Hypothesis
The first step in this process is to know what your experimental hypothesis is. The experimental hypothesis will drive questions like:
- What subsets are going to be examined? a. Antigens to be analyzed.
b. Suggested gating strategy. - What assay should be performed? Examples are: a. Phenotyping.
b. Cell cycle.
c. Proliferation. - What data needs to be extracted? a. Expression of a specific antigen.
b. Changes in population levels.
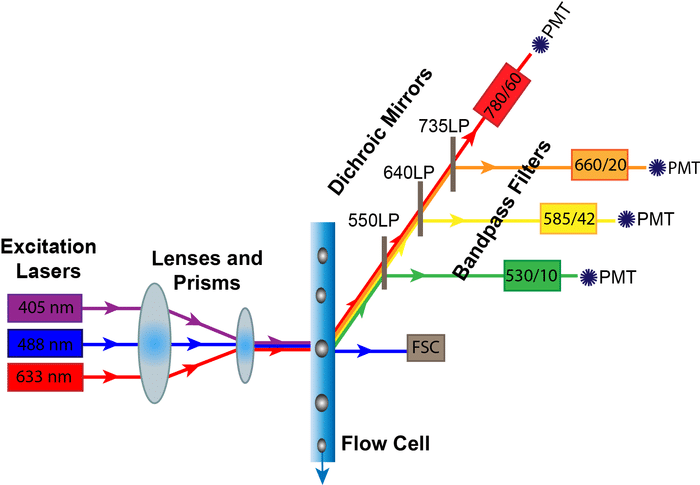
Step 2: Get to Know Your Instrument
The second step is to know a bit about the instrument you’re going to be using. This is critical to know what fluorochromes are possible candidates to use. Coupled with knowing what the instrument is capable is measuring, it is critical to know the relative brightness of different fluorochromes. There are several different places on the web that this can be found, including this article from Bitesize Bio on Choosing the Right Fluorochromes for Your Flow Cytometry.
Step 3: Find out the Relative Expression Level of Your Antigens
A third part of the equation is to know the relative expression level of the antigens to be measured. This step requires some Google-fu to building the list of available antibodies and fluorochromes. This is critical to building the panel (step 4), and can be time consuming. There are tools out there that can facilitate this such as Fluorish, Chromocyte and Fluorofinder. Each of these tools has their strengths and weaknesses, but all will help with the time consuming process of determining what fluorochrome/antigen combinations are available.
Step 4: Building Your Panel
Now comes the fun part – building the draft panel(s). In essence, a sudoku puzzle must be solved. The following rules apply to the puzzle:
- Identify regents that can’t be changed (such as fluorescent proteins, reactive oxygen reporters, proliferation dyes).
- Pair highly expressed antigens (e.g. CD3, CD4, CD20) with dimmer flurochromes.
- Pair the antigen(s) of interest with the brightest flurorochromes.
- Don’t forget dump channel and viability dye (but don’t design around them).
- Minimize compensation issues.
Dump channels and viability dyes are essential elements in any polychromatic flow cytometry panel. A dump channel is one where you place antigens that identify cells in the sample, but not of interest. These can be coupled in a single channel and when it comes to analyzing the data, the cells that are negative for the dump channel fluorochrome are carried forward for analysis. The dump channel helps clean up the analysis by removing cells that may contaminate downstream analysis. For example, in the case of the identification of murine hematopoietic stem cells investigators will add lineage markers (B220, CD3, Ter119, GR-1, CD11b) in one channel and exclude those from further analysis.
The use of viability dyes cannot be underemphasized. Since dead cells will non-specifically take up all antibodies in the panel at different rates, they can mimic the cells of interest. Including a viability dye is essential to reduce this background.
In both cases develop the panel first before adding these two critical stains. There is sufficient diversity in the viability dyes that they can be slotted anywhere. Likewise, consider using biotin for your dump channel antigens, so that you can come back with a streptavidin labeled conjugate in the appropriate color.
Step 5: Optimizing Your Panel
This may result in several different possible panels. To determine the optimal panel leads us to step five – optimization of the panel. Rarely does the first panel work out, so it’s critical to optimize the reagents and learn from each draft panel to create a final panel. The optimization includes optimizing signal to noise of each reagent in the panel by titration and voltage optimization, identifying the correct controls and ensuring the populations of interest are identified.
This outline will help you design your flow panel – be it four colors or fourteen. It is about the balance between what the instrument can measure, the expression level of the antigens, and the brightness of the fluorochromes. With that balance in mind, go forth and discover.
You made it to the end—nice work! If you’re the kind of scientist who likes figuring things out without wasting half a day on trial and error, you’ll love our newsletter. Get 3 quick reads a week, packed with hard-won lab wisdom. Join FREE here.
Put this article into practice
Choose a free resource to help you move forward
DOWNLOAD
Blood Collection Tube Chart
CHECKLIST
Journal Club Checklist