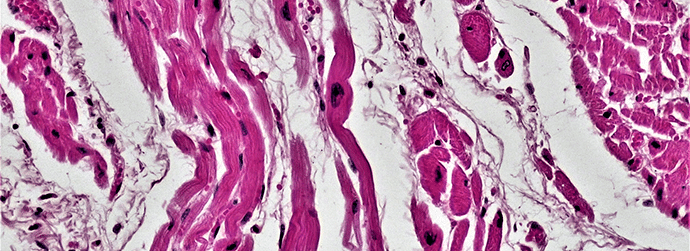
Haematoxylin and Eosin staining is the most common staining in the modern (and old!) histology lab. This staining technique gives an overview of the structure of the tissue and can be used in pathological diagnosis.
This article follows on from Nicola’s introduction, but we’ll take an in-depth look at the stains, chemistry and method to enable you to perform this to high standards in your own lab.
A colorful corner
You’ll hear Haematoxylin and Eosin referred to as ‘H and E’ or ‘HE’ staining (we’ll use H and E). If you are lucky enough to work in an institute which has a dedicated histology/microscopy lab, then they will probably have a dedicated H and E workstation. Just have a look for the very colorful corner of the lab- the workstation will either be in a fumehood, or on top of a downflow bench.
Lakes of dye
Haematoxylin is a natural dye which is extracted by boiling the logwood tree which is found in South America and West India. This extracted Haematoxylin is not actually a very good dye- for use in histology, it is combined with a metallic salt. This metallic salt is called a ‘mordant’- basically, it helps the haematoxylin to bind to bind to tissue. The most common metals used are aluminium and iron. If you hear of ‘haematoxylin lakes’, fear not! These are not vast Dead Sea-like bodies of dye- the term ‘lake’ just means a solution of haematoxylin with a metal salt (and some additional ingredients, but we’ll cover the composition of the H and E solutions in a forthcoming article).
Put this article into practice
Choose a free resource to help you move forward
POSTER
Immunofluorescence Troubleshooting Guide
POSTER
Fluorescent Proteins Guide
It’s all blue
Haematoxylin is used to dye cell nuclei. It binds to histones via the mordant. The slides to be stained are usually submerged in the solution for five minutes. This relatively long time ensures that all available binding sites are saturated.
When you take your slides out of the haematoxylin solution, nearly everything will be blue (including the staining rack)! At this stage, the nuclei will be deep purple/blue/red, but the next stage will render the stained nuclei dark blue, but the colour can be adjusted by repeating certain steps in the method.
Bluing cells
There are two stages used to achieve blue nuclei. The first is called ‘differentiation’- the slides are placed in an acid-alcohol solution which removes the excess staining. However, the remaining haematoxylin bound to the nuclei will still be a very dark and deep purple colour. To achieve the classic H and E blue colour, the slides are then rinsed in tap water followed by a step called ‘bluing’. This is done in a solution generally known as ‘Scotts Tap Water’.
Eosin next
The next stage is where the eosin comes in. This counterstain is used to stain cytoplasm, muscle fibres, etc. This pink counterstain also helps to differentiate between nuclei and non-nuclear components in cells. It is important not to over-stain at this stage as this will result in tissue/cells which are not immediately easy to decipher in terms of parts of the cell.
Very, erm, ‘-ish’!
The most commonly used eosin in histology labs is ‘eosin Y’. The ‘Y’ in this case stands for ‘Yellowish’. That’s right ‘-ish’!! Not yellow, but sort of yellow. Even though it stains pink. Eosin Y is a derivative of fluorescein. The other type of eosin you may encounter is ‘eosin B’. Can you guess what the ‘B’ stands for? Well, it’s ‘Blueish’!! The mechanism of eosin staining is not fully understood, but thought to be electrostatic in nature.
The slides are usually in the eosin for one to two minutes before being rinsed in tap water. It’s best to err on the side of caution and check your sections as you go instead of leaving them in too long, although you can remove a certain amount of overstain by placing the slides in a container with running water.
H and E Method
Below is a basic protocol for H and E staining in which I have split into three parts to include the dehydration and rehydration steps assuming you will be starting with a paraffin section and going through to mounting in synthetic medium.
Dehydration
|
Step |
Solution |
Time |
Notes |
| 1 | Xylene | 5 mins | All steps up to 10 should be carried out in a fume hood or downflow bench |
| 2 | Xylene | 5 mins | |
| 3 | Xylene | 5 mins | Drain off excess xylene before (4) |
| 4 | Absolute ethanol | 20 secs | |
| 5 | Absolute ethanol | 20 secs | |
| 6 | Absolute ethanol | 20 secs | |
| 7 | 95% ethanol | 20 secs | |
| 8 | 80% ethanol | 20 secs | |
| 9 | 70% ethanol | 20 secs | |
| 10 | Tap water | 2 mins | Running tap water in a sink |
H and E Staining
|
Step |
Solution |
Time |
Notes |
| 1 | Harris’ Haematoxylin | 5 mins | Run control slides first to check times and staining. |
| 2 | Tap water | 20 secs | Running tap water in a sink |
| 3 | 1% Acid alcohol | 5 secs | 5 secs ismaximumtime in this solution |
| 4 | Scotts Tap Water | 2 mins | Until tissue sections turn blue |
| 5 | Eosin | 2 mins | |
| 6 | Tap water | 20 secs | Running tap water in a sink, then check slides. |
Once you have achieved the desired staining, you will need to dehydrate your slides again before mounting.
The nuclei within your tissue should be dark blue, cytoplasm will be pink. If present, then erythrocytes are red, fibrin and muscle are deep pink and collagen stains pale pink.
This is simply a reverse of the Dehydration table above. Start with the 70% ethanol in step 9 and work back to step 1. You should allow excess fluid to drain back into the corresponding staining dish before proceeding to the next step.
Slides should be mounted directly from the last xylene incubation. We take a look at slide preparation in a different article, but check back here next week and we’ll explain the materials, methods and top tips for the solutions used in H and E.
You made it to the end—nice work! If you’re the kind of scientist who likes figuring things out without wasting half a day on trial and error, you’ll love our newsletter. Get 3 quick reads a week, packed with hard-won lab wisdom. Join FREE here.
Put this article into practice
Choose a free resource to help you move forward
POSTER
Histological Stains Poster
POSTER
Fluorescent Proteins Guide