Histology is the study of tissues and their microscopic structures. It plays a crucial role in biological research, aiding our understanding of cellular organization and pathology.
Tissue staining is a fundamental technique used in histology to enhance contrast and visualize cellular and tissue components. Among the various staining methods, hematoxylin and eosin staining is probably the most widely used.
In this article, we delve into the discovery of hematoxylin and eosin (H&E) staining, explore its mechanism of action, and discuss the structures it effectively stains.
Why We Need to Stain Tissues
Once a tissue specimen has been processed by a histology lab, and transferred onto a glass slide, it needs to be appropriately stained for microscopic evaluation. This is because unstained tissue lacks contrast: all of the fixed materials have a similar refractive index and a similar color. If you viewed an unstained tissue section under the microscope, everything would appear a uniform dull grey color.
Enjoying this article? Get hard-won lab wisdom like this delivered to your inbox 3x a week.

Join over 65,000 fellow researchers saving time, reducing stress, and seeing their experiments succeed. Unsubscribe anytime.
Next issue goes out tomorrow; don’t miss it.
The staining process therefore makes use of various dyes that stain particular cell components within tissues, so that you can distinguish different cell parts from each other.
A Very Short History of Hematoxylin and Eosin
For routine examination, H&E is the stain of choice. It adds color to otherwise transparent tissue sections so that you can get a detailed view of the tissue structure under a microscope.
As its name suggests, H&E stain is a combination of two dyes—hematoxylin and eosin.
The two stains were independently introduced in 1865 and 1875, respectively, by Böhmer and Fischer. In 1876, Wissowzky described their use in combination as a tissue staining method for staining structures in different colors.
Despite its simplicity, this stain has stood the test of time. Even now, over a century later, H&E remains among the most frequently used tissue stains worldwide.
How Hematoxylin and Eosin Staining Works
Ionic bonding is the most important type of bonding in histologic staining techniques. It exploits electrostatic attraction between ions of opposite charge, one of which is fixed in the tissue and the second of which is in the dye.
Hematoxylin and eosin do not produce color randomly. Instead, the dyes exploit differences in the chemistry of the tissue to differentially color various tissue components.
Haematoxylin alone is not technically a dye, and will not directly stain tissues. It needs to be combined with a mordant—a compound that helps it link to the tissue. The mordant is typically a metal cation, such as aluminum.
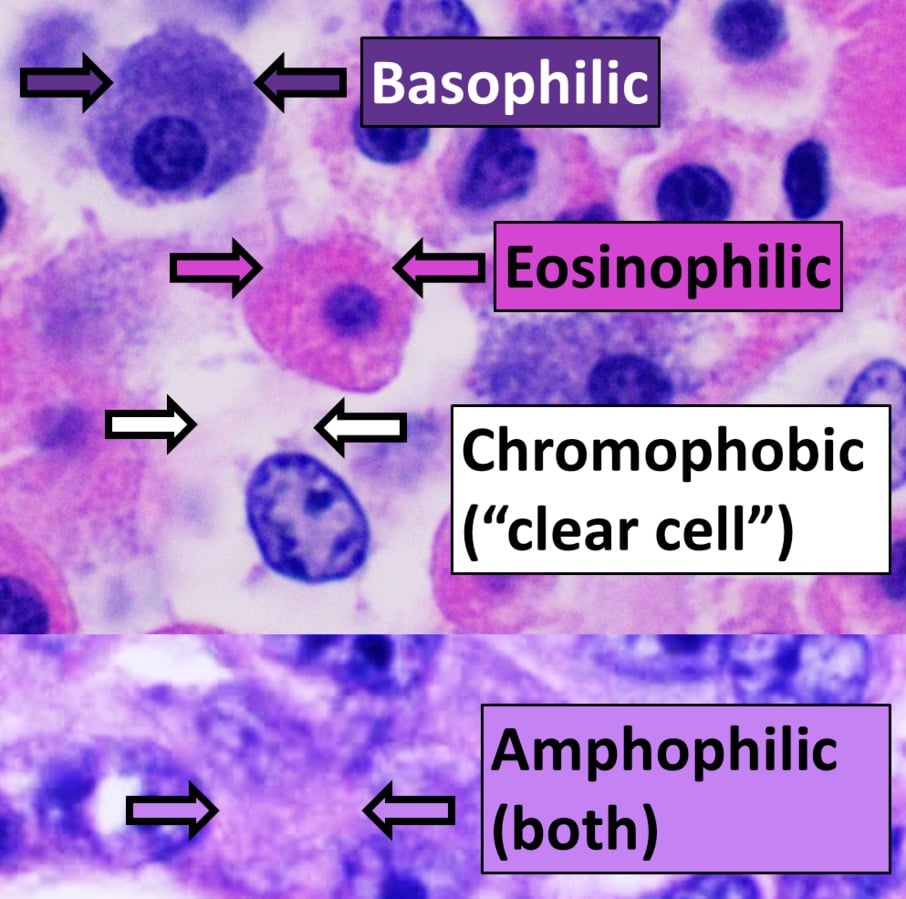
Hematoxylin in complex with aluminum is cationic and acts as a basic dye. Because it is positively charged, it can bind to negatively charged, basophilic cell components, such as nucleic acids in the nucleus. These get stained blue or purple as a result.
Eosin is anionic and acts as an acidic dye. It is negatively charged and can bind to positively charged, acidophilic components in the tissue, such as amino groups on proteins in the cytoplasm. These are typically stained in various shades of pink.
Structures You Can Stain Using H&E and Their Color
So what, specifically, can you stain using H&E? In a nutshell, here’s what H&E can stain and the colors produced:
- Nuclei: Hematoxylin stains cell nuclei blue or purple.
- Ribosomes: Hematoxylin stains ribosomes blue or purple.
- Cytoplasm: Eosin stains the cytoplasm shades of pink or red.
- Collagen and extracellular matrix: Eosin stains collagen fibers pink or red.
Check out Figure 1 below for an example of a tissue section stained using H&E.
Structures You Cannot Stain Using H&E
What can’t you see using H&E? What won’t it stain? Because H&E stain is ionic, it’s poor at staining most neutral components, including:
- Lipids.
- Glycogen.
- Mucus.
If you need to stain these structures and are wondering which stain to pick, check out the free poster at the end of this article.
Seeing Things Clearly: H&E Staining Summarized
Hematoxylin and eosin staining is a useful all-purpose method that is quick and easy to do, explaining why it has stood the test of time. It’s excellent for staining charged structures but less useful for neutral ones.
What are your tips for tissue section staining? Leave them in the comments section below! And check out our Histology Hub for all your tissue analysis needs.
Originally published in July 2012. Revised and updated in August 2015. Revised and updated again in July 2023.
Download Your Free Histological Stains Poster
If you want to go beyond hematoxylin and eosin staining, why not download our free histological stains poster and brighten up your lab? It lists common stains, the color they stain tissues, and some useful notes! Get your free copy here.
You made it to the end—nice work! If you’re the kind of scientist who likes figuring things out without wasting half a day on trial and error, you’ll love our newsletter. Get 3 quick reads a week, packed with hard-won lab wisdom. Join FREE here.








