According to the International Society for Stereology, the area of scientific study encompassed by this term is that which analyzes solids. If that all sounds a bit too much like materials science, then for us microscopists, it’s really about the review of three-dimensional objects (mainly tissues) by making horizontal and vertical incisions.
Stereology can be used to estimate cell population, determine cell size, regional volume, area fraction and surface area. Stereology is a complex practice. It involves many variables such as the ‘dissector principle’, ‘physical dissector, variation analyses and ‘guard volume’.
Stereology provides quantitative data and tends to provide unbiased results, but as with all types of scientific research, biases and inaccuracies do occur, the primary reason for repeating any study.
Methodology
Before setting out to do stereology, the researcher needs to acquire data such as the length, surface area, and volume (which is usually done within the stereology program you’ll be using).
Put this article into practice
Choose a free resource to help you move forward
POSTER
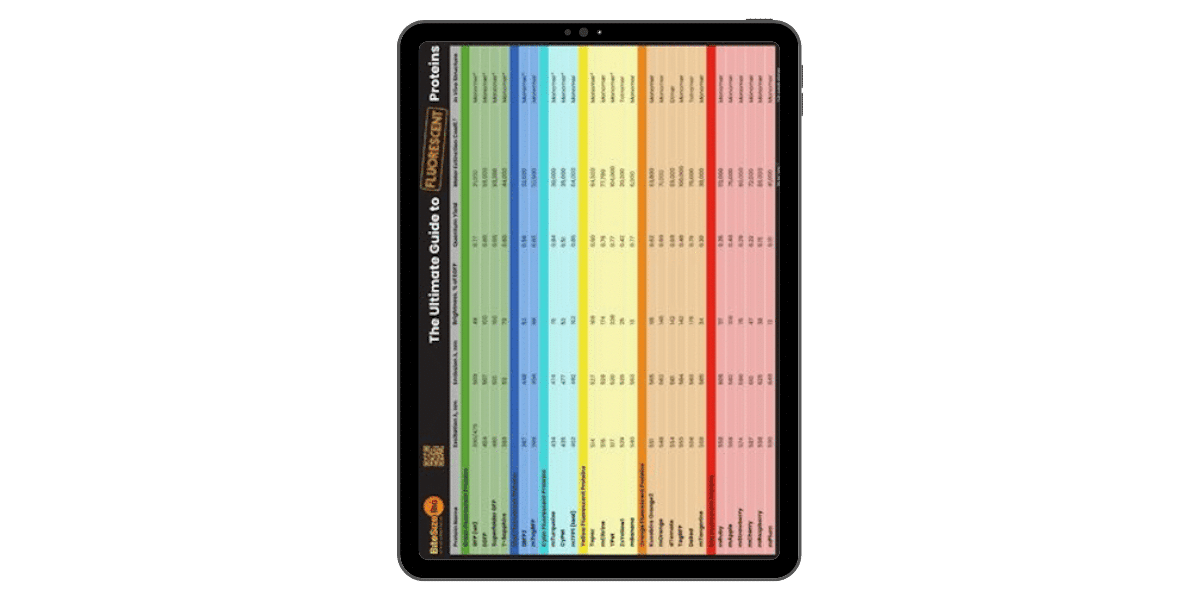
Fluorescent Proteins Guide
POSTER
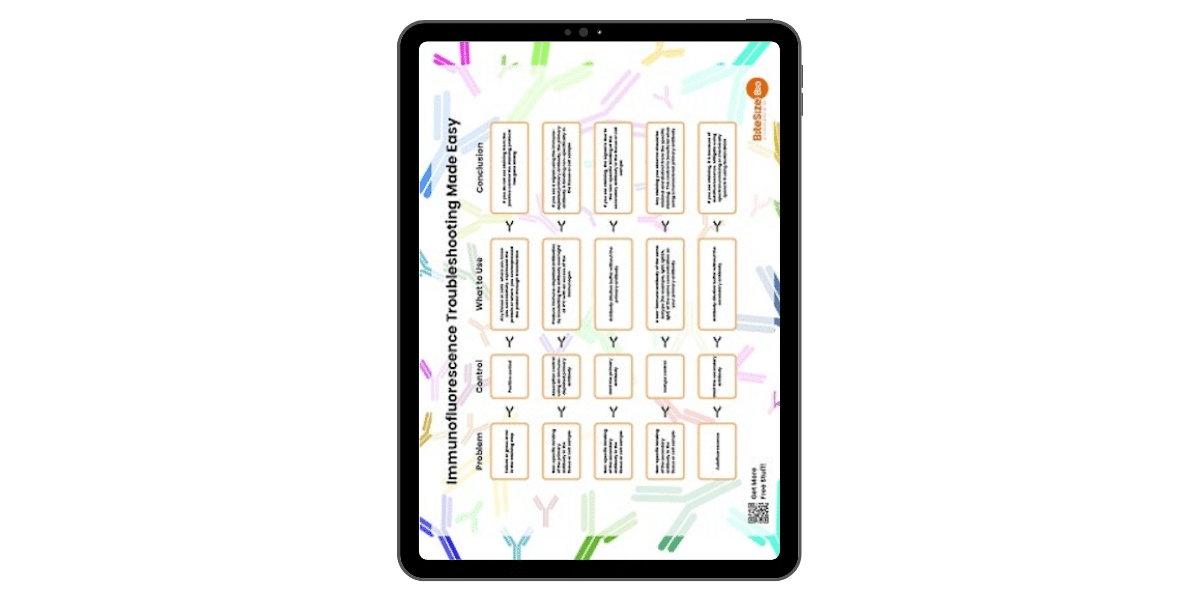
Immunofluorescence Troubleshooting Guide
Additionally, it is necessary to set certain criteria for the sample so that the most specific and accurate data can be acquired. Naturally, being able to identify and distinguish cells of interest from other cell types is of primary importance! After establishing these basics, a hypothesis can be developed and your theory tested. For example, this can range from the percentage of connective tissue within a tumour to the size range of blood vessels within a muscle biopsy.
Unbiased Stereology
When conducting any test (not just stereology) we should always be aiming for the most accurate results. To attain this goal, a researcher must perform unbiased analysis. To establish a bias-free environment for stereology testing, there must be complete access to the material in question. It is also important to avoid using any sample if it does not meet pre-established criteria.
Using certain principles, it’s possible to make efficient and accurate estimates of the number of cells in a well defined biological structure. In particular, the ‘optical dissector’ method can be used for counting cells in thick sections of tissue.
Each cell of interest should have a unique point visible in order to be counted, however, cell shape, size, orientation or distribution should not be a factor. An unbiased counting frame is widely used in many stereology programs and the counting rules are established within this frame. If cells are within the frame, then they are counted. If the lines of the frame fall upon a cell, then you should establish counting criteria which you should use for all of your analysis. For example- count any cells upon which the bottom and left hand side lines of the counting frame falls. Discount those cells which fall under the lines of the top and right hand side.
Fractionated Sampling
A key component to conducting an unbiased study is to randomly choose the population to provide testing samples, also referred to as ‘fractionated sampling’. The entire structure must be included in the available sections, be uniformly stained and with a tissue thickness of typically >20 µm. The presence of clear structural boundaries also needs to be taken into account. Of course, you’ll need to determine how much needs to be counted to get a good estimate- this is called the ‘coefficient of error’ (CE). The CE within your sample should be below 15%.
Once you are aware of all the factors involved in developing the methodology, then you can analyse your samples using a variety of tools and techniques to give unbiased and accurate results.
You made it to the end—nice work! If you’re the kind of scientist who likes figuring things out without wasting half a day on trial and error, you’ll love our newsletter. Get 3 quick reads a week, packed with hard-won lab wisdom. Join FREE here.
Put this article into practice
Choose a free resource to help you move forward
POSTER
Histological Stains Poster
POSTER
Fluorescent Proteins Guide