Safety Comes First: Common Laboratory Signs

Our laboratories are full of signs, aren’t they? From the chemical bottles to the basins, signs are everywhere. But that’s a good thing and important for our safety.
The lab safety signs on bottles, for example, tell you about the properties of the product you’re about to use so that you can take appropriate precautions while using it. You can also use these signs to label bottles after you’ve made a new solution to let others know what it is.
I remember my first days in the lab—lost in the signs. It took me some time to figure out the details of all these signs, so I’ve pulled together a quick overview of the most common lab safety signs to help you out.
Flammable Material
This is probably one of those self-explanatory signs—chemicals labeled with this are flammable and you should store them accordingly. Some laboratories have separate storage facilities for such chemicals (e.g. we have special security storage cabinets outside the lab for storing large bottles).
When using flammable chemicals make sure you keep them away from any oxidizing substances, flames, or sparks. You should also wear eye protection when working with highly flammable substances. Some examples of flammable chemicals we regularly use in the lab are ethanol and isopropanol for plasmid preparation.
Oxidizing Material
These substances are responsible for removing an electron from another substrate and are thus one component involved in an oxidation–reduction reaction. You should be very careful involving them in chemical reactions (e.g. mixing them with another chemical) while preparing a solution.
Oxidizing agents usually transfer oxygen to another chemical substrate. In this way, they provide oxygen to flammable substances to burn when used in the lab. You should always store them separately from flammable substances!
Oxidizing agents may also set you and your clothing on fire if you’re not careful—so don’t forget your gloves, eye protection, and lab coat as precautions.
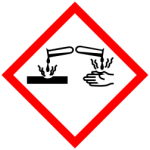
Corrosive Material
These are strong chemicals that can corrode your skin or other substances. There are many chemicals in our daily laboratory life that belong to this category.
Strong acid (e.g. sulfuric acid) and strong alkali (e.g. sodium hydroxide) solutions are both corrosive. The \beta-mercaptoethanol you use for reducing the disulfide bonds of your protein before running an SDS-PAGE or the crystal violet dye you use for staining your bacteria are also corrosive (along with their other individual hazards).
One drop of these corrosive substances can cause you serious eye damage! When working with corrosive substances, non-corrosive gloves, eye protection, and lab coats are all essential.
You should also make sure you know where the eye-wash station is, in the event of any accidents.
Toxic Material

These are highly harmful substances and, in extreme cases, can even cause death if you swallow, inhale, or absorb them through your skin.
Examples of toxic chemicals include the HCl you use for adjusting your buffer to the right pH and the universal pH indicator methyl orange. You should always use eye protection, gloves, and a face mask to prevent inhalation when working with toxic substances. And don’t forget: handle the chemical inside a fume cupboard.
Irritant
These substances can irritate your eyes and skin, causing itchiness, soreness, redness, and blistering. They can also cause toxicity if you inhale or swallow them.
For example, the calcium chloride you use to make your competent cells and the SDS for your protein gels are both irritants. You should be careful while preparing solutions containing irritants and make sure you protect yourself properly.
Health Hazard
Chemicals in this category can cause you serious health damage, including reproductive toxicity, problems with your respiratory system, germ cell mutagenicity, and carcinogenicity. Many of the chemicals that you are using carry such serious health hazards, so make sure you know this lab safety sign!
For example, the ethidium bromide used in DNA agarose gels is a potential mutagen. Phenol and chloroform both belong to this category too—phenol is a reproductive toxin and its vapor is also corrosive to the eyes, skin, and respiratory tract. Acrylamide is carcinogenic and neurotoxic too.
So make sure you use appropriate PPE—eye protection, nitrile gloves (for chloroform you need special 8 mil or heavier nitrile gloves), a lab coat, and a face mask are all essential when working with such substances.
Environmental Hazard
These chemicals are potentially hazardous to the environment; if not properly disposed of, they can contaminate soil and water and can be lethal for aquatic animals and trees. You should be very careful while disposing of these substances!
For example, bromoform and phenol are environmental hazards. Before you start working with these chemicals in the lab, make sure you learn the rules for disposing of them.
Explosives and Compressed Gas
Explosives are not generally seen in most labs, but compressed gas, on the other hand, is a fairly common sight (e.g. CO2 cylinders in tissue culture).
Gas cylinders and aerosol cans are compressed gases that should be treated with caution. Stocks of compressed gas cylinders are normally stored separately from the main lab in special safety cabinets.
Biohazard
This sign can be found on the doors or trash cans of your lab so that you are aware that you are entering an area in which biological materials, such as cell lines, bacterial or human samples, are used. The signs also indicate where to discard waste associated with your organisms.
For example, cloning regions of interest into a plasmid using E. coli is a standard laboratory technique, but imagine what could happen if we let all of these cloned bacteria free in our environment! Or worse—maybe you are working with contagious organisms; we could get in serious trouble if these organisms left the lab.
Protect yourself properly when working with such organisms, avoid any direct contact, and make sure you know what bins you should use for disposal.
Laser and Ionizing Radiation

These lab safety signs generally appear only in areas where you should have a fairly intensive safety introduction before beginning work.
Lasers can harm your eyes, causing severe injury; you should be well protected before entering labs that use lasers.
If you have to work with radiolabeled isotopes (e.g. you want to run an EMSA), you generally have to check with your doctor first that you’re in a fit condition for such work. Make sure you’re fully informed about all the safety rules and the type of PPE you’re required to use to protect yourself and those you work with before your enter these labs.
High Voltage

Be it your PCR product or your expressed protein, you want to see whether or not it is the right product and the most common (and probably most cost-effective) method for visualizing this is running your product through a gel using an electric field.
When running gels, be careful! If you’re not paying attention you can end up giving yourself an electric shock. You can find the high voltage sign in your gel electrophoresis apparatus, so don’t forget to switch off the main switch before your take your gel out of your running buffer.
Hot and Cold Temperatures
Extreme temperatures are another potential lab hazard.
I think most labs have one electric plate or induction cooker for sterilizing small quantities of solutions, or for creating a supersaturated solution if you’re struggling to dissolve something. Some of these hot plates can reach temperatures of 450°C, so be careful you don’t give yourself a nasty burn when using them!
At the other end of the spectrum, it’s also important to understand the safety concerns associated with extremely cold temperatures; the –80°C freezer is the standard storage option for cDNA and glycerol stocks, and how often do you use liquid nitrogen to freeze your samples?
Special cold-resistant PPE is required to use both the –80°C and liquid nitrogen facilities to avoid frostbite.
Lab Safety Signs Summarized
While this list of lab safety signs isn’t exhaustive, and you’ll have to look up the other warning signs present in your lab, there are a few general rules you can follow to keep yourself safe in the lab. It’s always best to wear:
- a long-sleeve lab coat (you can always fold your sleeves when you work in the laminar airflow bench);
- full pants (yes, even on those hot summer days when your lab air conditioning isn’t working);
- closed shoes;
- eye protection.
Make sure you know what type of chemicals or hazards you will be working with and protect yourself accordingly—prevention is better than cure! Check out our article on how to carry out a risk assessment.
Are there any lab safety signs that you think we should add? Let us know in the comments.
Want to make hazard symbols clear to everyone to keep your lab and buddies safe? Download our free lab safety poster and pin it up in your lab.
Originally published July 18, 2016. Reviewed and updated August 2021.











oxygen,carbon monoxide,concentrated glucose solution & concentrated sodium chloride solution… Which of them in corrosive item and which of them in explosive item.. can you reply my comment quickly? I really2 need your answer ASAP.. Thanks
some are wrong .
Hi Imran,
Which ones and what location are you from? Maybe it’s a geographic thing? Thank you!