If you’ve ever measured a metabolic readout and thought, “That’s interesting… but I’m not entirely sure what it actually means,” you’re not alone.
At the bench, metabolism is often approached one pathway at a time. You measure glucose uptake, lactate production, ATP levels, or a single metabolite, and then infer what the cell is doing energetically. The problem is that cells don’t experience metabolism as isolated pathways. They experience metabolism as a tightly connected network, where changes in one metabolite often reflect shifts in how cells allocate resources.
A more helpful way to think about metabolism, especially when interpreting cellular metabolism assays, is to view it as a network of metabolic crossroads. Metabolic crossroads are points in metabolism where multiple pathways intersect, and where relatively small changes can signal much larger shifts in cellular state.
In this article, we’ll look at five metabolites that sit at critical metabolic crossroads and explore how they can provide powerful insights into cellular metabolism in health and disease. These are:
- Pyruvate
- Malate
- Glycogen
- branched-chain amino acids (BCAAs)
- β-hydroxybutyrate (BHB)
Pyruvate: Where the Cell Decides What Happens Next
We often describe pyruvate as the end product of glycolysis, but from an experimental perspective, that description is misleading. Pyruvate sits at one of the busiest metabolic crossroads in the cell, linking cytosolic glucose breakdown to mitochondrial energy production and biosynthesis.
Once formed, pyruvate can be converted into acetyl-CoA to fuel the TCA cycle and ATP generation, reduced to lactate to regenerate NAD⁺ and sustain glycolysis, or diverted into biosynthetic and gluconeogenic pathways. Which route the cell chooses depends on oxygen availability, mitochondrial capacity, redox pressure, and growth demands.
Because pyruvate feeds into multiple downstream pathways, changes in your pyruvate-related readouts rarely reflect glycolysis alone. For example:
- In cancer cells, elevated pyruvate flux often supports aerobic glycolysis and biosynthesis, rather than efficient ATP production.
- In non-proliferative or energy-limited contexts, altered pyruvate handling may instead point to mitochondrial dysfunction or redox imbalance.
When pyruvate levels change, the most helpful question is not “did glycolysis increase?” but “where is the cell sending this carbon, and why?”.
That shift in thinking is central to accurately interpreting metabolic readouts.
Malate: Reading Mitochondrial Health and Redox Balance
Malate rarely attracts much attention, but it plays a quietly central role in metabolic interpretation. It sits at a crossroads between mitochondrial energy production and cytosolic redox balance.
Malate participates in both the TCA cycle and the malate–aspartate shuttle, allowing reducing equivalents generated in the cytosol to be transferred into mitochondria to maximise ATP yield from glycolysis. Because malate has a dual role, changes in its levels provide insight into how well mitochondria cope with energy demand and redox stress.
In cancer cells, particularly those growing in hypoxic environments, malate metabolism frequently reprograms to support altered TCA flux, NADPH generation, and biosynthetic needs rather than maximal ATP production. Therefore, malate can act as a sensitive indicator of metabolic rewiring.
If your experimental question involves mitochondrial function, oxidative stress, or adaptive metabolism, malate often tells you more than ATP alone, provided you analyse it in the context of other metabolic pathways.
Glycogen: A Readout of Metabolic Flexibility, Not Just Fuel Availability
Researchers often treat Glycogen as a simple indicator of how much glucose a cell has access to. Experimentally, however, glycogen levels tell you far more about metabolic flexibility—how well a cell can buffer energy fluctuations and adapt to changing conditions over time.
When glycogen levels fall, it’s easy to conclude that cells are glucose-starved.
But your reduced glycogen measurements can just as easily reflect increased energetic demand, stress adaptation, or a strategic shift toward alternative fuels, particularly if other metabolic readouts remain stable.
Because glycogen integrates signals from glucose uptake, hormonal regulation, and energy demand, it is best to interpret changes in glycogen content alongside metabolites such as pyruvate and BHB.
In the context of these additional metabolites, glycogen becomes a powerful indicator of whether cells are metabolically adaptable (or locked into inflexible energy-use patterns), rather than a simple measure of glucose exposure.
BCAAs: Where Metabolism Meets Signaling
BCAAs, which comprise leucine, isoleucine, and valine, occupy a unique position at the intersection of metabolism and signaling.
While BCAAs can serve as energy substrates or building blocks for protein synthesis, changes in BCAA levels often reflect how cells sense and respond to nutrient availability, rather than simply indicating shifts in amino acid supply.
For example, altered BCAA levels can indicate changes in nutrient signaling pathways such as mTOR activation, shifts in growth prioritization, or altered metabolic state, even when overall energy production remains unchanged.
Viewed alongside metabolites such as pyruvate and glycogen, BCAAs help reveal whether cells are prioritizing growth, maintenance, or stress adaptation. In this way, BCAAs act less like passive metabolic intermediates and more like reporters of cellular decision-making in response to nutrient cues.
BHB: Evidence of a Metabolic State Shift
BHB is the most abundant ketone body, but it is also a signaling metabolite with wide-reaching effects. Although BHB is often measured as a marker of fatty acid oxidation or ketosis, rising BHB levels usually signal a broader metabolic state change rather than an increase in a single pathway.
Elevated BHB typically reflects a coordinated shift away from glucose-dependent metabolism toward alternative fuel use and signaling pathways that change cellular priorities. This transition may occur in response to nutrient limitation, energetic stress, or disease-associated metabolic rewiring. Interpreting BHB in isolation can therefore be misleading, particularly if it is assumed to represent increased fat metabolism alone.
When evaluated alongside metabolites such as glycogen and pyruvate, BHB helps clarify whether cells are undergoing a broader metabolic shift, such as adapting to limited carbohydrate availability or energy stress, rather than simply increasing fatty acid oxidation.
Worked Example: Why Single Metabolite Readouts Can Mislead
Imagine you measure elevated pyruvate in a cell-based experiment. Taken alone, it’s tempting to conclude that glycolysis has increased. However, when you look at other metabolic crossroads, the interpretation can change.
If you also find an elevation in malate and a reduction in glycogen stores, the data may instead reflect the rerouting of carbon toward biosynthesis and redox support rather than increased ATP production. In this context, elevated pyruvate points to metabolic rewiring, not simply “more glycolysis”.
Now let’s consider glycogen depletion. On its own, low glycogen might suggest glucose starvation. Still, if BHB levels are rising at the same time, the cells may have shifted toward alternative fuel use, indicating metabolic adaptation rather than energy failure.
These examples highlight why interpreting cellular metabolism assays based on a single, isolated readout can be misleading.
Meaning emerges when you examine how multiple metabolites at key metabolic crossroads change together under the same experimental conditions.
Interpreting Cellular Metabolism Assays With Confidence
Cell metabolism underpins cellular health and function, and disruptions to metabolic pathways are implicated in a wide range of diseases and pathologies. Understanding cellular metabolism depends not just on what you measure (Table 1), but also on how you obtain those measurements.
A strategic, multi-metabolite approach is essential for confident evaluation of metabolism. Measuring metabolites that sit at key metabolic crossroads allows you to distinguish between superficially similar outcomes, like increased glycolysis versus metabolic rerouting, or energy stress versus adaptive fuel switching, using evidence rather than assumption.
Table 1. Roles and Rationale for Monitoring Key Metabolites.
| Metabolite | Primary Roles in Metabolism | Why Monitor It? |
| Pyruvate | • Links glycolysis to TCA cycle • Supports biosynthesis and redox balance | • Reveals shifts between aerobic and anaerobic metabolism • Indicator of mitochondrial function and cancer-associated reprogramming |
| Malate | • TCA cycle intermediate • Redox shuttlingNADPH generation | • Reports on mitochondrial health, redox balance, and anaplerotic capacity |
| Glycogen | • Glucose storage • Rapid energy mobilization | • Reflects hormonal regulation, energy availability, and defects in glucose handling |
| BCAAs | • Protein synthesis • Energy substrate • Metabolic signaling | • Biomarker for muscle metabolism, insulin sensitivity, and nutrient signaling balance |
| BHB | • Alternative fuel • Signaling metabolite | • Indicates metabolic state, ketosis, stress adaptation, and inflammatory modulation |
Promega’s bioluminescent metabolic activity assays support this interpretation-driven measurement strategy. By enabling sensitive, quantitative analysis of metabolites such as pyruvate, malate, glycogen, BCAAs, and BHB, these assays allow you to assess how multiple pathways shift together under the same experimental conditions (Figure 1).
Rather than providing isolated readouts, this assay portfolio supports the deliberate selection of complementary measurements to help you build a coherent picture of how cells allocate energy, carbon, and reducing power in response to perturbations.
Because the assays are ready-to-use and compatible with a wide range of sample types, including suspension and adherent cells, organoids, tissue homogenates, serum, and plasma, they can be applied consistently across experimental models without adding unnecessary technical complexity.
By choosing assays that reflect metabolic context rather than single pathways, you move from collecting metabolic data to interpreting metabolic behavior with confidence.
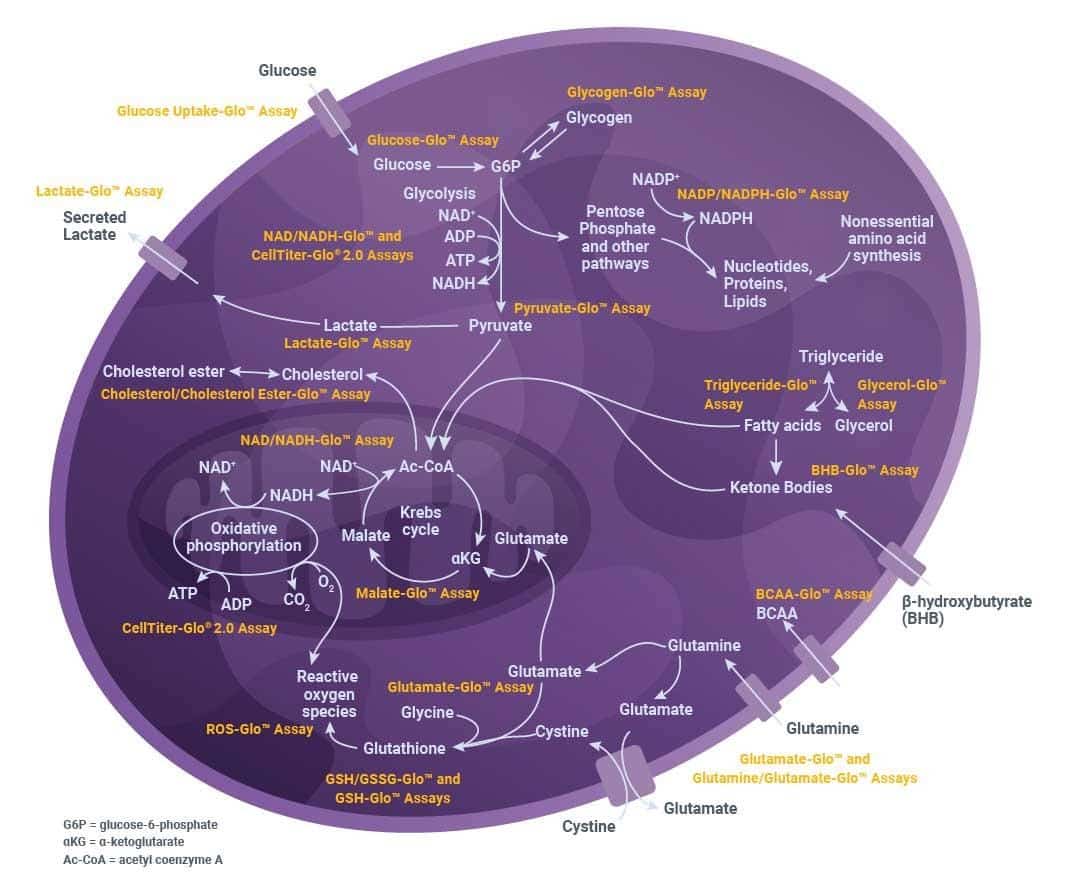
Figure 1. Evaluating metabolic pathways. Major cellular metabolic pathways form an interconnected network that can be analyzed using a range of assays.
Conclusion: Moving From Measurements to Meaning
Metabolic data only becomes useful when you can place it in the context of what the cell is trying to do. Focusing on metabolites that sit at metabolic crossroads helps you see how energy, carbon, and reducing power are being allocated, rather than assuming that a single pathway has increased or decreased in activity based on a single, isolated readout.
In practice, taking a network-based approach to interpreting cellular metabolism assays makes it easier to distinguish genuine metabolic shifts from misleading snapshots, leading to conclusions that are more reproducible and biologically meaningful.
Sources
Moe, S. (2025) The Greatness of Glycogen: A Central Storage Molecule in Energy Metabolism – Promega Connections.
Moe, S. (2025) The Power of Pyruvate, A Pivotal Player in Cellular Energy Metabolism – Promega Connections.
Haupt, K. (2024) The Marvel of Malate: A Crucial Component in Cellular Energy Metabolism – Promega Connections.
Moe, S. (2025) The Benefits of BCAAs: Branched-Chain Amino Acids in Health and Disease – Promega Connections.
Moe, S. (2025) The Brilliance of BHB: A Key Ketone Body in Metabolic Health – Promega Connections.








