You’ve nurtured your cells for weeks, perfected your experimental conditions, and nailed down all the controls. You’ve harvested your cells and gently lysed them, now you’re ready to look at the proteins. What’s one of the most common next step in protein analysis? A denaturing gel or SDS-Polyacrylamide Gel Electrophoresis!
SDS-Polyacrylamide Gel Electrophoresis, or SDS-PAGE for short, is the technique where proteins are denatured and linearized, then run across a current through a thin gel, which separates the proteins by size. SDS-PAGE is a key step in many experiments including:
- Western Blots
- Separating proteins for mass spectrometry
- Determining protein purity (such as after an immunoprecipitation or protein purification)
- Examining protein size
- Quantifying protein abundance
- Visualizing post-translational modifications
…. Basically just about any experiment where you need to know something about your favorite proteins. SDS-PAGE is an essential lab tool you’ll want to master. And part of mastering it is understanding how it works.
Unraveling the Proteins
The first step in running a denaturing gel is to denature your proteins. This is accomplished using:
Enjoying this article? Get hard-won lab wisdom like this delivered to your inbox 3x a week.

Join over 65,000 fellow researchers saving time, reducing stress, and seeing their experiments succeed. Unsubscribe anytime.
Next issue goes out tomorrow; don’t miss it.
SDS
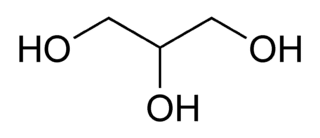
When you have your proteins in hand — whether they are from a cell lysate or purified sample — denaturing your proteins is the first step and for this you need Sodium dodecyl sulfate (SDS). SDS is the main star of the denaturing protein gel. SDS is a detergent composed of a hydrophobic hydrocarbon tail attached to an ionic sulphate group and a key component of loading buffer,. When SDS meets up with your protein, SDS’s hydrocarbon tail dissolves any hydrophobic region of the protein, while the sulfate end breaks non-covalent ionic bonds. This causes your protein to lose its secondary and tertiary structure, and well…unfold. Once surrounded by SDS, your previously carefully folded protein becomes loose and long, just like overcooked spaghetti.
A Reducing Agent
Also in the denaturing mix is the noxious ?-mercaptoethanol (?-me). Its job is to break disulphide bonds, which further spaghettifies your protein. Its strong smell also lets everyone else in the lab know you’re doing a protein gel!
Heat
The final factor in denaturing your protein is heat. Typically, you will boil your protein samples in the loading buffer (containing Tris-HCl, SDS, bromophenol blue, glycerol, and ?-me) before loading them in your gel. This helps to completely denature the proteins and also helps with physically loading the gel. Protein samples frequently are gummy, particularly if the protein prep is from cell or tissue extracts and therefore contains DNA. Boiling homogenates your sample, as the heat melts any DNA in the prep, in turn making it less gummy and easier to load on the gel.
Separating the Proteins
Now that your proteins are nice and linear, it’s time to run them out on your polyacrylamide gel using electricity. That’s right it is time for PolyAcrylamide Gel Electrophoresis or PAGE.
The Resolving Gel
The main component of PAGE is the resolving acrylamide gel. Acrylamide gels are composed of SDS, buffer, ammonium persulfate, and TEMED. This mixture is poured between two closely spaced plates, forming a thin gel. Once polymerized this mix creates a sieve-like network that the proteins can then travel through. As the proteins run through the gel, they will work through the mesh structure of the gel. Larger proteins take longer to navigate through the gel, while smaller proteins move faster. Therefore, you can adjust how much proteins will separate by changing the percentage of acrylamide in the gel. Less acrylamide means a gel with larger pores, which good for large proteins. More acrylamide means a gel with smaller pores, which is great for separating smaller proteins.
The Current
The proteins don’t move through the gel by their own volition. Instead you must subject your gel to an electric current, with the negative charge at the top where the proteins are loaded and the positive charge at the bottom. SDS-coated proteins have a large negative charge (thanks to the SDS), thus the proteins are attracted to the positive charge and move from top to bottom.
The Stacking Gel
A stacking gel is poured at the top of the gel. The stacking gel is made of the same stuff as the resolving gel but with a lower concentration of acrylamide. It is poured, after your resolving gel polymerizes, immediately before loading the gel. This is where you place your loading comb, which will create neat wells for your protein sample. A decent stacking gel is important to ensure crisp, sharp bands on your gel. The stacking gel ensures that, regardless of protein size and sample volume, all your proteins enter the gel at the same time.
A Few Cautions
Heat is a friend and foe.
There are two points during an SDS-PAGE where you need to keep an eye on the temperature. First, is boiling your sample. Larger proteins may need a longer boiling time to facilitate denaturing, while smaller proteins may degrade with too much heat. Therefore, it’s a good idea to test your boiling time when using a new protein sample. The second heat issue is during the gel run. Running a current across the gel generates heat. Running the gel too fast, at too high of a current, can over-heat your gel causing it to warp or even melt. This will not make for a pretty gel, so try to avoid cranking up the current because you want to go to lunch.
Birds of a Feather Flock Together
Proteins of nearly the same size will migrate at the same rate. If you are looking for two similarly sized proteins, or trying to visualize small post-translational modifications, you may need to tweak the acrylamide percentage up for better resolution, or run your sample longer.
And there you have SDS-PAGE! When your gel is finished running, carefully separate the plates and move on to the next step of your experiment!
You made it to the end—nice work! If you’re the kind of scientist who likes figuring things out without wasting half a day on trial and error, you’ll love our newsletter. Get 3 quick reads a week, packed with hard-won lab wisdom. Join FREE here.