Your advisor tells you that he wants you to use HPLC to analyze your compound. You know you’ve heard of this technique before, but you can’t remember what HPLC stands for, let alone how to go about doing it! We’ve all been there, and I bet you wish you had paid more attention in that lecture!
Fear not – in this series of articles I will remind you about the power of the HPLC column :)
In this first article, I will take you through the principle behind HPLC and remind you of its uses – you will be ready for the lab in no time!
How does HPLC work?
High-performance liquid chromatography, or HPLC, is a long name for a powerful technique based on the simple fact that individual compounds behave differently in water.
HPLC separates and purifies compounds according to their polarity, or their tendency to like or dislike water. To put polarity into context, consider that oil is an apolar liquid that doesn’t mix with water. Ethanol, on the other hand, is polar and as many of you know, mixes very well with water (Vodka and coke anyone??).
I have tried to simplify the whole process in Figure 1 below, but first let’s look at the main components involved in HPLC. Apologies in advance for some unavoidable jargon!
The components
The HPLC column
This is also known as the stationary phase. This is the workhorse of the HPLC machine, is made from one of a variety of substances (often silica) and is very compact in nature. The silica particles are functionalized by long carbon chains. The carbon chains are apolar and therefore the longer the chain, the more apolar the column becomes. Columns containing 18-carbon chains are commonly used and are known as C18 columns.
The HPLC sample
Sample types vary greatly depending on the field and the type of compounds in question. HPLC can be used to analyze compounds in biological specimens (urine, blood, saliva, and muscle), environmental samples, medicinal chemistry (drugs), and microbiology (toxins produced by fungi and bacteria).
Injection of sample
Samples are injected into the HPLC column. This used to be carried out manually, meaning that some poor soul had to sit by the HPLC machine for hours on end injecting each sample with a syringe, sometimes all night long!
Luckily, newer models have an automatic injector, reducing the manual input and allowing higher throughput. Modern machines are equipped with software allowing the user to input a list of samples, how much and in what order they should be injected. So you can enjoy your lunch while your HPLC runs itself!
The mobile phase
This is really just a mixture of water and an organic solvent (usually acetonitrile or methanol). The mobile phase gets it name because it moves through the column and at the same time elutes (or flushes out) the compounds from the column.
Compounds are often eluted along a concentration gradient. If you’re anything like me, concentration and gradient are two words you hate to see coming together in a sentence! It just means that the percentage of water in the mobile phase decreases over time, while the percentage of the apolar solvent increases simultaneously. This means that the mobile phase gradually becomes more apolar. Don’t worry too much about gradients for now, as they will appear again in a follow-up article.
The HPLC run
HPLC can be performed in a number of modes. The most commonly used method is known as reversed-phase (RP-HPLC) and this is what I describe here. In this mode, compounds are separated starting with the most polar, and ending with the apolar compounds. For all modes, a high-powered pump moves the sample and the mobile phase through the column. A typical run can take between 10-60 minutes.
The principle behind HPLC – a closer look
Now that you have an idea of the components involved, let’s move on to the principle in a little more detail.
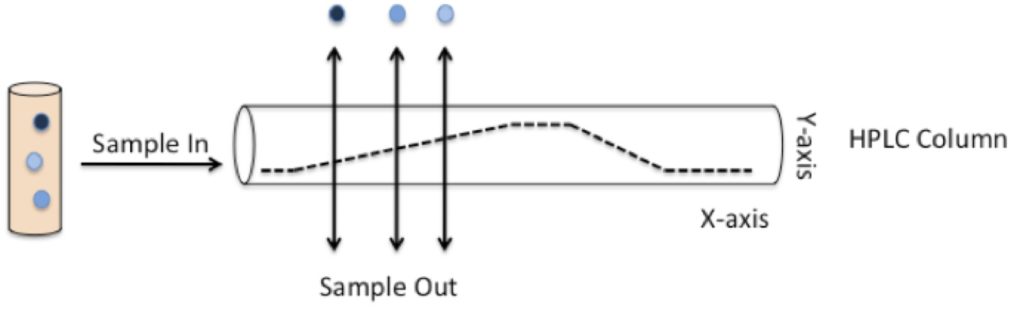
I mentioned above that HPLC separates compounds on the basis of polarity. But how does this actually work? English please! As the gradient kicks in, the solvent concentration increases while the water concentration decreases. This makes the mobile phase more and more apolar. Compounds contained in the sample will stick to the carbon chains in the column, with the most apolar compounds sticking the strongest, and the most polar compounds sticking weakly.
Figure 1 shows what happens to a sample containing a mixture of compounds after injection into the column. The compounds bind to the column and are flushed out at different times, depending on whether they are more likely to stick to the column or the mobile phase as it is pumped through. The time that each compound elutes (or flushes out) from the column is known as that compound’s retention time (Rf).
Figure 1: The principle behind HPLC
Understanding the output
The output or results of an HPLC run is usually viewed as a chromatogram (Figure 2). This is a horizontal series of peaks representing compounds eluted from the column with different Rf values. Modern HPLC equipment is often coupled to a diode array detector (DAD), allowing the user to look at the resulting chromatogram of separated compounds in wavelengths from 190 nm to 900 nm. If the compounds under investigation are known, the user can choose to look only at 1 or a few selected wavelengths. For instance, cocaine can be observed at 254 nm.
Figure 2: A typical HPLC chromatogram

Applications
Within biology and medicine, HPLC is often used as an analytical tool to assay biological and environmental samples for the presence or absence of known compounds (for example metabolites, drugs, toxins, pesticides), and can assist in the identification of unknown compounds.
Within chemistry, however, HPLC is routinely used to monitor chemical reactions, as well as to determine the purity of the products. In addition, the process of HPLC can be modified to preparative HPLC, whereby compounds of interest can be purified for further use.
HPLC can come across as being very complicated to begin with, but rest assured, just like most other lab techniques, it makes a lot more sense when you actually do it.
Stay tuned for my follow-up articles in the coming weeks, in which I will go through some pointers on how you can get the best from your HPLC run depending on your research goal, as well as discuss objectively how HPLC can be used within your research.
Are there any other aspects of this technique you would like covered in more detail? If so, we would love to hear from you!
Happy HPLC-ing :)