Welcome to the last article in the series on E. coli origins of replication where we will touch upon plasmid incompatibility! This is a broad and complicated topic so we will mainly focus on points that might be relevant for you in the lab.
What is Plasmid Incompatibility?
Plasmid incompatibility is usually defined as the failure of two co-resident plasmids to be stably inherited together in the absence of external selection.1 In simpler terms, if the introduction of a second plasmid negatively effects the inheritance of the first, the two are considered to be incompatible.
Plasmids can be seen as selfish entities in evolutionary terms. Having gained territory in a bacterial cell, they will try to prevent any other plasmid co-residing with them. In other words, they want the bacterial cell all for themselves! Plasmid incompatibility is a very effective strategy for protecting territory within the bacterial cell.
As we’ve discussed throughout this series, the number of plasmids in a cell is governed by elements encoded within the origin of replication (ori). It is not possible to maintain two different plasmids that use the same mechanism for replication in a single cell. Therefore, plasmids fall into compatibility groups base on their replication strategy and you cannot use two plasmids in the same cell system if the plasmids belong to the same compatibility group. Incompatibility groupings are a popular way of classifying plasmids and the current list of groupings can be found in a recent review.2
Do I Need to Worry About Plasmid Incompatibility?
Generally, incompatibility only becomes an issue if your work requires that two plasmids be maintained together, so before you start out a big cloning experiment where you require two plasmids make sure they don’t belong to the same group!
The good news is that while plasmids from the same group cannot reside together, there is usually no effect on the survival of two different groups in a single cell. This is because replicons from different compatibility groups do not usually interact with each other in the cell.
What Makes Plasmids Incompatible with Each Other?
Competition for Replication Factors
It is widely accepted that competition for replication factors leads to competition between plasmids. Plasmids with inherent growth advantages such as faster replication (due to their smaller size) or less toxicity have the potential to rapidly outgrow other plasmids in the cell. This is even more likely to occur when the plasmid copy number is low. Taking into account the number of cell division cycles occurring in a single overnight culture, even small differences in competitive advantage are thought to be able to lead to rapid dominance of a culture by a single plasmid.
This is Linked with Copy Number
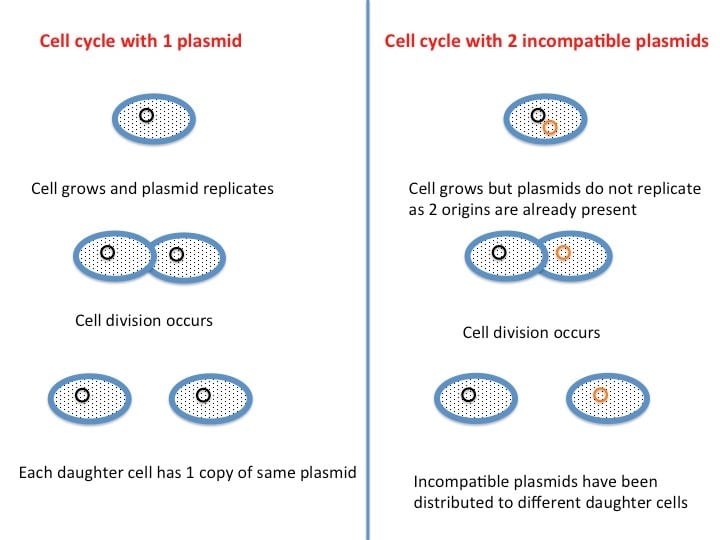
Plasmid incompatibility is intrinsically linked with copy number. What does this mean? When a cell contains two incompatible plasmids, the cell cannot distinguish between the origins at the point of replication initiation. The result is that replication is postponed until after the two plasmids have been distributed to different cells to restore the pre-replication copy number as illustrated in Figure 1.
Figure 1. Copy number and plasmid incompatibility are linked.
Incompatible plasmids cannot be distinguished from each other at the point of replication initiation, and are therefore distributed to different daughter cells. Figure adapated from genes.atspace.org.3
Types of Plasmid Incompatibility
Different categories of incompatibility exist and these can be summarized as symmetric or vectorial incompatibility. During symmetric incompatibility, the co-resident plasmids are equally likely to be lost while vectorial incompatibility stipulates that one plasmid is lost exclusively or with higher probability than the other. These categories are discussed extensively in a review by Novek et al.1
Is There Are Way Around Plasmid Incompatibility in the Lab?
It is possible to identify mutations that reduce or eliminate incompatibility between two plasmids by selecting plasmids of the same group for their ability to coexist. This can be done in the lab if both plasmids have different selectable markers, e.g. one with ampicillin resistance and one with hygromycin resistance. Incompatibility mutations are probably best studied in the ColE1 plasmid family, and an extensive review of this topic can be found elsewhere.4
So that was it for this short series about E. coli origins of replication. Thanks for reading to the end! Hopefully these articles have given you some understanding about this vast and complex topic and you have gained some new information that can help you plan your experiments in the lab. Do you have any tricks for working with incompatible plasmids? Please feel free to get in touch if you would like to share your experience with us.
References:
- Novick RP, Clowes RC, Cohen SN, Curtiss R, Datta N, and Falkow S. (1976). Uniform nomenclature for bacterial plasmids: a proposal. Bacteriol. Rev. 40:168–89.
- Carattoli A. (2009). Resistance Plasmid Families in Enterobacteriaceae. Antimicrob Agents Chemother. 53(6):2227–38.
- GENES IV: The replicon: 12.15 Plasmid incompatibility is connected with copy number.
- Camps M. 2010. Modulation of ColE1-like Plasmid Replication for Recombinant Gene Expression. Recent Pat DNA Gene Seq. 4(1):58–73.