Brightfield Illumination
This defines the most basic method of optical microscopy using white light to illuminate the sample in the transmitted mode. Absorption and diffraction of the light by the molecules in the specimen generates the contrast in the image. Methods such as darkfield illumination, differential interference contrast and phase contrast help to increase the contrast in the resulting image.
Fluorescence
Specific fluorescent molecules, called ‘fluorophores’, can be excited by light and absorb photons at specific wavelengths. This absorption switches the fluorescent molecule into the excited state, which is followed by the return to the ground state by emitting a photon of a longer wavelength. The time between the absorption of an excitation photon and the emission of a photon is in the range of nanoseconds and this ‘fluorescence lifetime’ is specific for a given fluorophore and its microenvironment. Fluorophores can run through thousands of excitation/emission cycles before they bleach and get converted into a dark state. Fluorophores can be designed, synthesised and coupled to functional molecules for very specific labelling in biological samples. Naturally occurring, genetically encoded fluorophores, so-called ‘fluorescent proteins’, have been modified for use in optical imaging and have greatly improved live specimen imaging.
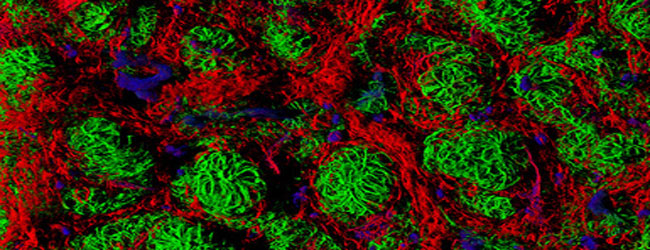
Fluorescence Microscopy
The use of this technique in biomedical research has literally revolutionised light microscopy. Fluorescent labelling has greatly increased the specificity and contrast in imaging. Using fluorophores precisely targeted to molecules or structures in a biological specimen, the illumination of the microscope specifically excites the fluorescent labels in the sample. The emitted light can be separated by filters according to its spectral properties and recorded by highly sensitive detectors such as photomultiplier tubes, photon counting diodes and CCD cameras. More on CCD cameras in the next part of this Glossary! The imaging of the bright fluorescent signal on a completely dark background delivers an excellent image contrast, and the use of several spectrally different fluorophores allows the synchronous detection of more than one species of molecule in the same sample.
Optical Filters
These are devices which selectively transmit, absorb or reflect light of different wavelengths. In a conventional configuration, these are pieces of optical glass with a special coating or a dye in the glass body that has absorbing or reflecting characteristics for very accurately filtering spectral bands. The general nomenclature for each of the optical filters reflects their spectral separating properties. For example, an ‘LP650’ is a Longpass filter with a ‘cut-on’ wavelength of 650 nm. This means the filter will absorb all frequencies of light below 650 nm and transmit light above 650 nm. Knowledge of the filter properties in the light path of your microscope is absolutely essential for correct image acquisition. Another example is a ‘BP500-520’ filter. This is a Bandpass filter that transmits a relatively narrow spectral wavelength range between 500 and 520 nm. The following abbreviations on the most commonly used filters refer to the given spectral characteristics, ‘KP’ and ‘SP’ for short pass, ‘BP’ for band pass, ‘LP’ for long pass, ‘ND’ for neutral density, and ‘BG’ (Blue glass) for red blocking filters.
Put this article into practice
Choose a free resource to help you move forward
POSTER
Fluorescent Proteins Guide
EBOOK
Guide to Special Stains for Histology
In Part 2, we will explain a bit about Dichroic Mirrors, Cameras, Photomultiplier Tubes and what is meant by ‘Confocality’.
You made it to the end—nice work! If you’re the kind of scientist who likes figuring things out without wasting half a day on trial and error, you’ll love our newsletter. Get 3 quick reads a week, packed with hard-won lab wisdom. Join FREE here.
Put this article into practice
Choose a free resource to help you move forward
POSTER
Immunofluorescence Troubleshooting Guide
EBOOK
Guide to Special Stains for Histology