Currently, there are more than 75 antibody drug conjugates (ADCs) in various stages of pre-clinical and clinical development. The combination of a targeted antibody coupled with a cytotoxic small-molecule drug (via a flexible linker) makes for a lethal and specific oncologic drug product. However, an ADC is a heterogeneous cocktail of molecules with a range of drug antibody ratios (DAR). The complexity of this combination and the distinctive manner in which each component behaves chemically, poses numerous analytical challenges.
Analytical Challenges to ADC Analysis
We’ll cover four major challenges to ADC analysis and techniques to successfully overcome them:
Levels of Free Drug
ADCs contain drugs that cannot be delivered by traditional means because of their high cytotoxicity, with IC50 in the picomolar range. Determining the concentration of unconjugated drug is critical to ensure the safety of the drug product. These free drugs can be difficult to detect due to their low levels, usually in the 1-10 ng/mL range. Liquid Chromatography-Multiple Reaction Monitoring/Mass Spectroscopy (LC-MRM/MS) has the necessary sensitivity and specificity to determine free drug down to 1 ng/mL or less.
Drug-To-Antibody Ratio (DAR)
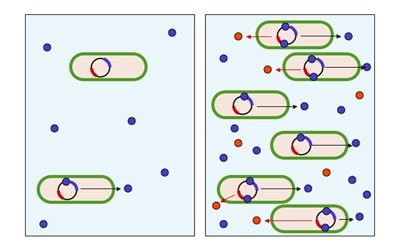
Conjugation of the monoclonal antibody and toxin typically results in a mixture ADCs with varied drug-to-antibody ratios. DAR is essential to the efficacy and, possibly, metabolism of ADCs and typically ranges from 2-8 drug molecules per monoclonal antibody. Determining the extent of conjugation, concentration, and distribution of these components is necessary for understanding and controlling the drug product and monitoring its stability. This ratio must be monitored during manufacturing, on release, and over time (stability studies). Hydrophobic interaction chromatography (HIC) with UV detection provides an excellent means of determining these characteristics.
Residual Solvents and/or Catalysts
You must bear in mind that conjugation is an organic chemistry process. Therefore, a number of organic solvents and inorganic compounds (including heavy metals) that are part of the conjugation must be detected and measured in the final drug product. These compounds are not typically part of a biopharmaceutical product, and many molecular biologists aren’t accustomed to the need to assess them for the presence heavy metals (e.g., palladium and platinum) and residual solvents . Inductively Coupled Plasma/MS (ICP/MS) has the required specificity and sensitivity for analysis of heavy metals. Residual solvents of interest include: dimethylamine (DMA), dimethylformamide (DMF) and THF (tetrahydrofuran). GC/MS is an excellent tool for this application, but requires some upfront sample clean-up.
Heavily Modified ADCs
The chemistry of antibody-drug conjugate linkers and toxins modifies the chemistry of the monoclonal antibody component of the conjugate. These modifications often result in increased antibody aggregation due to changes to the surface of the antibody protein. Aggregation decreases the efficacy of the entire ADC population and its overall stability. These aggregation events occur more often with ADCs than with typical monoclonal antibodies. Microfluidic imaging (MFI) is the industry standard technique for detecting sub-visible molecules (i.e., aggregation) and is utilized for in-process, release and stability testing.
Use the Right Tools for ADC Characterization
Critical issues for ADC characterization are target site-specificity, binding properties, stability of the linker and drug, toxicity/potency of the drug, free drug concentrations, conjugation sites, DAR, heterogeneity and aggregation. Under International Conference of Harmonization (ICH) guidelines for developing ADCs, testing will be necessary for identity, purity, impurities, activity, concentration and stability. Meeting these needs requires knowledge of both chemical and biological properties. But the right tools, used correctly, will prove to be up to the task.