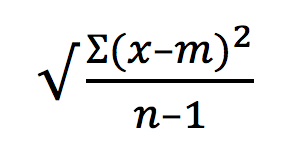One of the much sought after question asked by many researchers worldwide is – “What is the gene expression profile of a single cell within a heterogenous pool of cells?” While mass cytometry is the current ‘hot’ methodology for single cell analysis, the good old flow cytometry can help us perform rapid analysis of single cells.
Given that proteins are the orchestrators of all cell functions, concurrent detection of proteins associated with surface, cytoplasmic and nuclear compartments deliver a powerful approach for testing many of our hypotheses. Flow cytometry can be used to detect as many as 18 proteins per cell at >10,000 cells per second. If you would like to use flow cytometry techniques in your experiments and are asking “How to”, then read on.
Antibody based detection techniques including flow cytometry are based on “know thyself and thine enemy” approach. In other words, we need antibodies that both recognize our protein of interest and are conjugated to a fluorochrome to determine protein expression. This task is made relatively easy if a commercially available antibody already exists in various fluorophore-conjugated forms.
The Outside-In Approach to Immunostaining
Detection of proteins on the cell surface is the first step in immunostaining. Remember to perform the entire surface staining procedure on ice to slow the cycling of antigens between cell membrane and cytoplasm. Also, you must conduct the procedure in the presence of Fc-blocking reagent to reduce non-specific staining of cells.
The next step is to take a peek inside the cell to identify the proteins on the inner leaflet of the plasma membrane, the cytoplasm, and the nucleus. This involves first fixing the cells to immobilize the antigen-antibody complex on the surface and cellular structures using cross-linking reagents. It is followed by permeabilization (i.e., punching holes in the cell membrane) to allow entry of antibodies inside the cell to bind and detect the protein. Always fix the cells first and then permeabilize the cells—otherwise the contents of the permeabilized cells might leak out and result in loss of your protein of interest.
The most commonly used fixatives are 4% Paraformaldehyde, 95% Ethanol, 100% methanol, and 100% acetone. The concentration, temperature, and duration of fixative treatment can be optimized to get the desired results. However, longer incubation times with fixatives sometimes result in masked or destroyed epitopes. Some fixatives, like methanol and acetone, can both fix and permeabilize the cells (time-saver!).
When an additional permeabilization step is needed, detergents like NP-40, Triton-X, and Saponin are routinely used. They provide access to antibodies by making large enough holes in both the plasma and nuclear membrane. As with the case of fixatives, the concentration and duration of permeabilization treatments does affect the staining pattern.
The Easy Way Out
As always, many commercially available kits are available for cell fixation and permeabilization. The choice of kit depends on the location of protein of interest inside the cell and the composition of the kit reagents. For example: BD Cytofix/Cytoperm Plus Fixation/Permeabilization kit is typically useful for intracellular cytokine staining. The Cytofix buffer contains formaldehyde as the fixative and the Perm/Wash buffer has the mild detergent, Saponin. It is important to remember that all the intracellular staining steps (e.g., anti-IFN? antibody) must be performed in the presence of Perm/Wash buffer to maintain the cells in a permeabilized state.
If you would like to stain for transcription factors and signaling molecules inside the nucleus, say Foxp3 or Bcl6, kits like the ebioscience Foxp3/Transcription factor buffer set work very well. These transcription factor-staining kits typically contain formaldehyde as the fixative along with a combination of a stronger detergent like Triton-X or methanol to attain accessibility through the nuclear membrane.
Tips & Tricks
- If you use FACS tubes to perform staining, the quantity of the buffers needed is large. One easy option is to use 96 well u or v bottom plates for staining.
- Following fixation, cells structures are cross-linked and so cycling of proteins is no longer a worry. Therefore, additional permeabilization and staining steps can be carried out at room temperature.
- Formaldehyde is a carcinogen, so utmost safety precautions should be taken while handling these buffers.
- Following fixation and permeabilization, cells become smaller in size and translucent. So, if you are not able to see your cell pellet, be assured you did not lose your cells!
- Because of the change in size of cells, higher voltage setting is needed when analyzing intracellularly stained cells on the flow cytometer. If you can’t find your cells on the flow cytometer, just crank up the voltages a bit. If you are unsure of the voltage settings, seek the help of your core facility manager.
- Commercially available fixable viability dyes can be used to eliminate background noise due to dead cell contamination.
- After fixation, cells can be resuspended in FACS buffer at 4ºC overnight in the dark and can be analyzed later on the flow cytometer following permeabilization and staining. However, there is a possibility that some antibodies might exhibit reduced staining intensity.
- Even though it is recommended to perform surface stain before fixation, some antigens, like CD4, can be stained concurrently with nuclear antigens, like Foxp3, after fixation/permeabilization. But this shortcut will only work for some antibody clones, so make sure you do a trial run.
- If your cells have reporter proteins like GFP in them, the fluorescence might be lost completely or diminished following use of buffers containing methanol. One solution is to use alternative buffers containing formaldehyde. One trick that worked for me is to use the BD cytofix/cytoperm first followed by treating the cells with ebioscience foxp3 staining kit for concurrent detection of fluorescent proteins and nuclear antigens like Foxp3.
In all, immunostaining involving cell fixation/permeabilization requires a lot of optimization/trial and error. Be assured, once you have mastered the technique for your individual needs there is no looking back. It opens the gates to the magical kingdom of cellular biology!
References:
- Bendall SC, Nolan GP, Roederer M, Chattopadhyay PK. A deep profiler’s guide to
cytometry. Trends Immunol. 2012 Jul;33(7):323-32. doi: 10.1016/j.it.2012.02.010. - Abcam. Fixation and permeabilization in IHC/ICC.
- Affymetrix: eBioscience. Staining Intracellular Antigens for Flow Cytometry.
- BD Biosciences. Optimizing Intracellular Flow Cytometry: Simultaneous Detection of Cytokines and Transcription Factors.