Filter plates simplify multi-step cell-based assays by allowing incubation, washing, and filtration to occur in the same well.
Filter plates vary in their design and materials. Choose the wrong plate for your system and it can undermine assay performance, leading to poor signal, inconsistent washing, or compatibility issues with automation.
So, how do you choose the right filter plates for cell-based assays, and know whether you need a different plate to enable automation?
In this article, you’ll find a five-step framework to help you select plates that support reliable and scalable automated workflows.
What Filter Plates Are and Why They Are Used
Filter plates are multiwell plates that have a microporous membrane at the base of each well, allowing liquid to pass through while retaining cells, proteins, or other assay components, depending on the membrane material and pore size.
Because the membrane acts as both a reaction surface and a separation interface, filter plates allow multiple assay steps to occur in the same well without transferring samples between vessels.
This format simplifies complex workflows and reduces handling variability, which is why filter plates are widely used in applications including ELISpot assays, cell migration, tumor invasion, cell membrane permeability, receptor-ligand binding, and drug transport studies.
Step 1: Define the Biological Capture Event at the Filter Membrane
Before any other variable is considered, spend a moment to clarify what physically needs to happen at the filter plate membrane to get your intended outcome. It could be cytokine adsorption, cell transit, ligand retention, or molecular size exclusion, for example. The mechanism determines the functional class of membrane required.
Consider a straightforward example. You want to measure interferon-gamma secretion from stimulated T cells. To do this, cytokine-specific capture antibodies must be immobilized on a solid surface, and that surface must hold cytokines locally around the secreting cell before they can diffuse away.
Now consider a migration assay, where the experimental question is whether a cell will traverse a defined physical barrier in response to a chemical gradient. Or consider a receptor–ligand binding assay, where the membrane must physically retain a receptor-bound fraction while allowing unbound material to pass through under vacuum or centrifugal force.
Three assays. Three biological questions that feel, at the highest level, like the same kind of inquiry: did the event happen?
But each depends on an entirely different membrane behavior: protein adsorption, mechanical transit, and selective physical retention.
A plate optimized for one will fall short in another.
The biological event being captured sets every downstream filter plate selection decision: the membrane chemistry required, the structural features that preserve signal integrity, and ultimately the automation compatibility of the plate… these all come later.
Step 2: Determine the Membrane Chemistry You Need
When choosing filter plates for cell-based assays such as ELISpot, high protein-binding membranes are required because secreted cytokines must be captured locally and held in place for detection (Figure 1).
Hydrophobic PVDF membranes (specifically Immobilon®-P) provide high protein and nucleic acid binding capacity at a 0.45 µm pore size in its standard ELISpot configuration. The white color of PVDF also creates a consistent optical backdrop that supports spot detection and quantification.
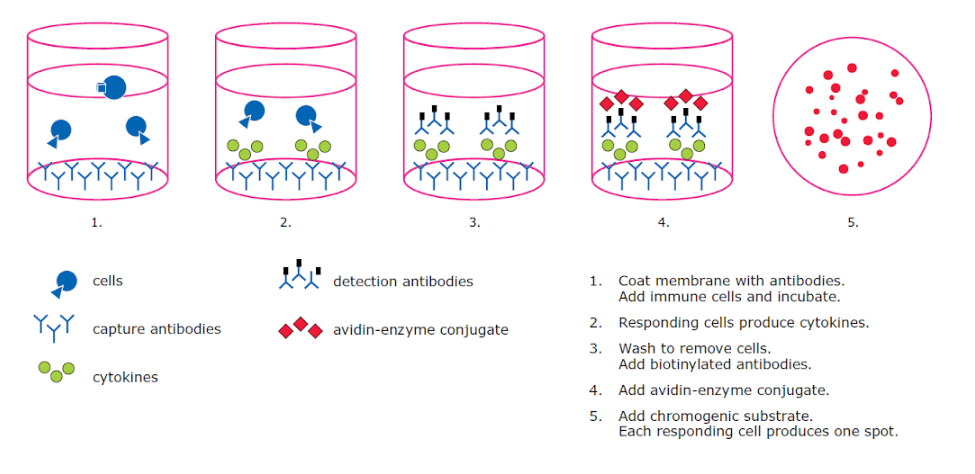
Figure 1. ELISpot workflow showing cytokine capture on a high-binding membrane.
Mixed cellulose ester (MCE) membranes offer similar high-binding characteristics and are used in comparable ELISpot contexts (though they typically have a lower binding capacity than PVDF membranes). In practice, these membranes are functioning as solid-phase capture surfaces: the cytokine must adsorb immediately and remain localized while the cell is alive and secreting.
The inverse requirement applies in solubility or sample preparation assays, where high membrane binding would cause drug compounds to be lost to the filter rather than collected in the filtrate. Here, low-binding membranes are appropriate. Hydrophilic PVDF (Durapore®) offers low protein and nucleic acid binding across a range of pore sizes from 0.22 to 1.2 µm, supporting sample preparation and biochemical assays where compound recovery is the priority.
Glass fiber filters occupy a mechanistically distinct category. In receptor-ligand binding assays, the filter retains the receptor and its bound ligand fraction during filtration-based separation, while the unbound ligand fraction passes through. This is not adsorption-based capture, it is physical retention by the filter matrix during rapid liquid passage. The distinction matters when selecting membranes for radiometric or binding-based formats, because the two mechanisms impose different requirements on membrane structure and flow dynamics.
Representing a categorical shift in mechanism, polycarbonate membranes do not bind or retain biological material—they provide a defined physical barrier through which cells either do or do not transit. These membranes are used in migration, invasion, and chemotaxis assays.
Choosing incorrectly across these filter plate membrane material categories can make a well-designed assay produce messy data.
Step 3: Confirm Appropriate Filter Pore Size Against Your Target Molecule and Mechanism
Once the membrane material is matched to the biological interaction you are trying to detect, pore size determines whether the physical event can actually occur at the intended scale and rate.
In cell migration and chemotaxis assays, pore diameter must be matched to the average diameter of the cells used. MultiScreen®-MIC (Migration, Invasion, Chemotaxis) plates are available in 3 µm, 5 µm, and 8 µm pore sizes to accommodate different cell types. These plates can support in vitroangiogenesis (tube formation) assays (Figure 2). Smaller pores restrict transit to cells that must actively deform to pass, while larger pores allow freer movement.
Selecting a pore too small produces mechanical resistance that suppresses the chemotaxis signal even when a genuine response is present; too large, and cells may pass non-specifically, obscuring the biological distinction between stimulated and unstimulated conditions.
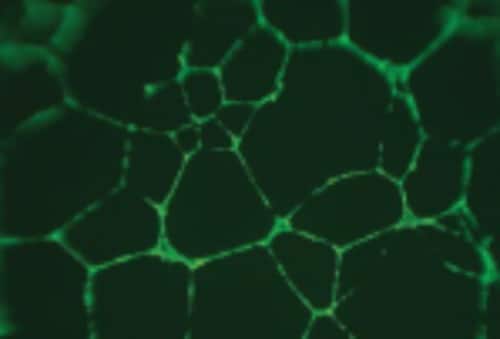
Figure 2. Angiogenesis (vessel formation) experiments were performed using HUVEC (human umbilical vein endothelial cells) on 5 µm MultiScreen®-MIC Filter Plates pre-coated with extracellular matrix. Cells were labeled with 8 µg/ml of Calcien AM fluorescent label.
For size-exclusion-based applications (e.g. PCR product purification and sequencing reaction cleanup), the membrane simply selects for a particular molecule size cut-off. Smaller molecules pass through while larger ones are retained, allowing rapid cleanup without chromatography.
Lysate-clearing applications follow a related logic to size-exclusion, where particulate material is separated from soluble components through the membrane. If pore size is poorly matched to the sample, contaminants may pass through or target material may be lost, compromising the purity of the preparation.
Flow dynamics also bear on assay reproducibility. In vacuum-driven filtration, pore size affects the resistance to flow across each well. You may need to verify that vacuum settings remain within the appropriate range for the specific membrane and pore combination being used, since flow uniformity across a 96-well plate becomes more difficult to maintain when resistance varies.
The governing question at this stage is the relationship between pore size and the biological entity being studied—cell, protein, or small molecule. Whether transit is intended to occur, be prevented, or be size-selective will determine which pore specification is appropriate, and it is this decision that connects membrane chemistry to the flow and automation constraints that follow.
Step 4: Consider Plate Architecture: How it Influences Isolation, Crosstalk, and Underdrain Needs
Membrane and pore selection define what the assay is physically capable of measuring. Plate architecture determines whether that measurement remains clean and reproducible across all wells.
Standard 96-well filter plates typically accommodate around 300 µL per well (or up to ~1.9 mL in deep-well formats).
In contrast, 384-well filter plates have much smaller wells (100 µL). These plates support miniaturized workflows that conserve reagents and precious cell samples. However, the smaller well volume and reduced surface area can make assay optimization more sensitive to factors such as washing efficiency, signal strength, and flow uniformity.
In practice, 96-well formats are often preferred during assay development or when signal intensity is limiting, while 384-well formats are adopted once workflows are stable and throughput becomes the primary constraint.
Well isolation is a primary structural requirement. In multiwell filter plates, individually sealed wells prevent incubation crosstalk between wells during the assay period.
This isolation matters most in ELISpot, where cytokine diffusion between wells during incubation would distort spot localization and edge-well counts, and in radiometric binding assays, where signal bleed across well boundaries adds background that cannot be subtracted with confidence.
Underdrain design contributes to both filtrate collection and membrane access. Recessed and removable underdrains (Figure 3) allow plates to stack securely during vacuum operations while also enabling direct access to the membrane surface when assays require post-filtration membrane staining or analysis—as in ELISpot visualization steps.
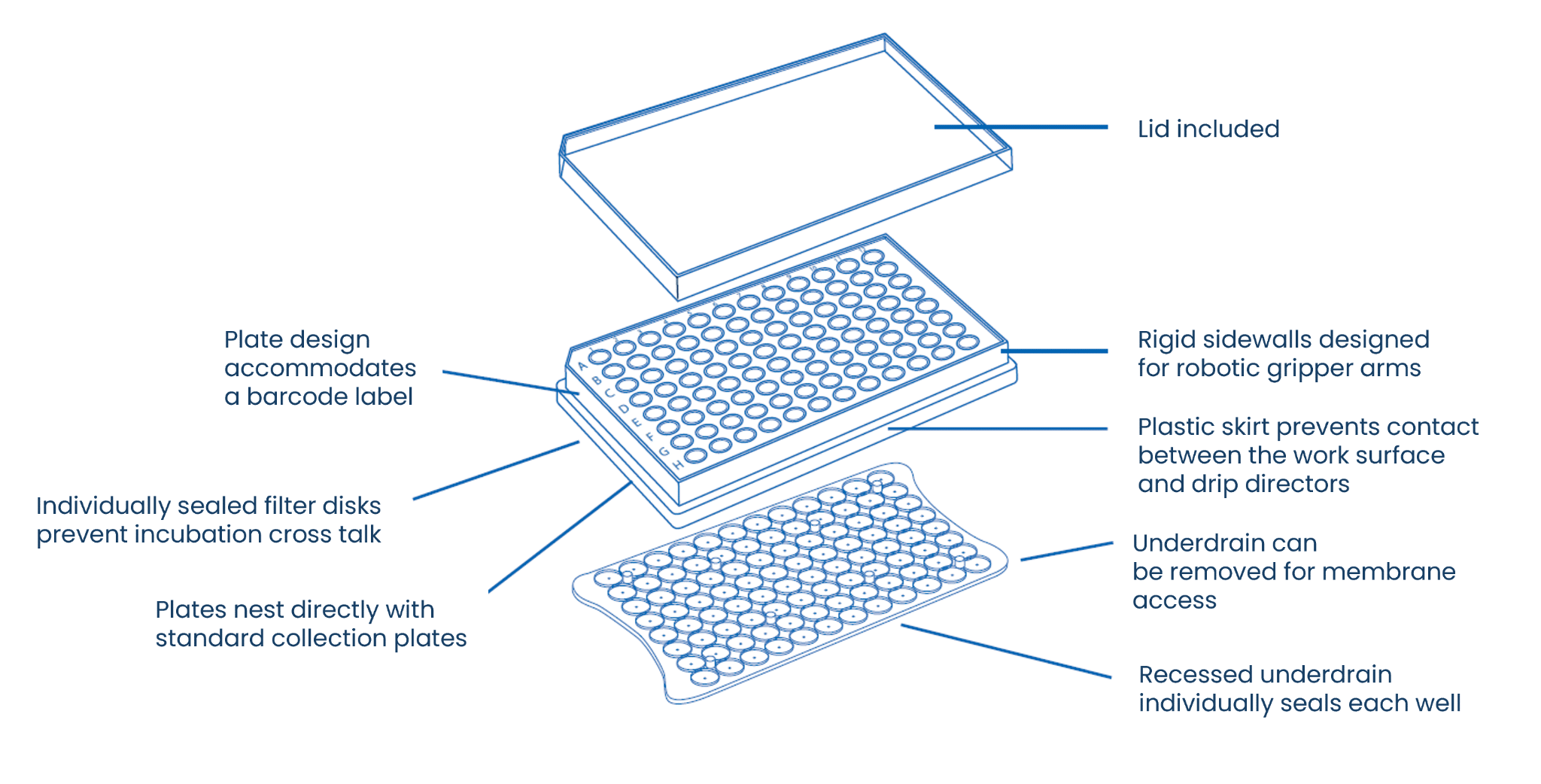
Figure 3. Key design features of the MultiScreen®HTS filter plate including rigid sidewalls for robotic handling, individually sealed wells to prevent crosstalk, barcode compatibility, and a removable recessed underdrain that allows membrane access and stacking with collection plates.
For assays where washing efficiency directly affects signal-to-noise, nylon mesh membrane backing provides structural reinforcement behind the membrane. In MultiScreen®HTS filter plates, this backing is designed to improve washing consistency across wells. A common issue in standard filter plates is that incomplete or uneven washing leaves residual signal that inflates background, particularly in outer wells where vacuum distribution can differ from central wells.
Step 5: Confirm Automation Constraints: ANSI/SBS Compliance and Flow Control
A filter plate that is scientifically correct for an assay will still fail in an automated workflow if its physical dimensions, handling characteristics, or flow behavior fall outside the tolerances that robotic systems require. Automation compatibility is a design requirement that must be confirmed before plates are introduced into automated decks.
Dimensional compliance with ANSI/SBS microplate standards ensures that plates are compatible with the robotic gripper arms, plate readers, and stacker systems found in high-throughput screening environments. Plates that deviate from these standards—even marginally—can cause grip errors or misalignment events that interrupt or invalidate entire plate runs.
Mechanical stability adds a separate constraint. Rigid sidewalls are a structural prerequisite for robotic handling: plates that flex under gripper pressure or deform during centrifugation introduce positional error that compounds across a run.
Flow control imposes a third layer of requirements. Recommended vacuum ranges for 96-well filter plates are defined and bounded. For MultiScreen®HTS plates, the recommended operating range is 8 in. Hg with a maximum of 18 in. Hg. Exceeding these limits can damage membranes or drive non-uniform flow across wells. Similarly, centrifugal protocols have defined force limits (1000 × g recommended, maximum 3000 × g) that must be respected to preserve membrane integrity and filtration uniformity.
For filtrate collection in vacuum-driven workflows, plate-on-plate stacking introduces the risk of filtrate cross-contamination between adjacent collection wells. DirectStack™ vacuum stacking technology addresses this contamination risk by enabling plate-on-plate stacking while minimizing crosstalk during filtrate collection.
In practice, automation compatibility must be confirmed at each of these levels—dimensional, mechanical, and flow-related—before assuming that a plate suitable for manual use will transfer directly to a robotic workflow without modification.
Building a Selection Framework: Biology → Membrane → Pore Size → Architecture → Automation
This five-step framework for choosing filter plates for cell-based assays applies across major application categories—ELISpot (standard and fluorescent), migration and invasion assays, receptor-ligand binding assays with glass fiber plates, and ADME (Absorption, Distribution, Metabolism, and Excretion) or solubility screening—without requiring a different logic for each.
The Millipore® MultiScreen® filter plate portfolio provides membrane options spanning high-binding PVDF, low-binding hydrophilic PVDF, glass fiber, polycarbonate, and mixed cellulose ester, across both 96- and 384-well formats to support assay miniaturization and throughput scaling.
Structural features—individually sealed wells, recessed and removable underdrains, ANSI/SBS-compliant dimensions, and rigid sidewalls—are incorporated to support both manual and automated workflows.
For assays at the intersection of multiple constraints, the MultiScreen® application matrix and associated technical resources can support membrane selection decisions.
References
- MultiScreen® Wall Chart (2025). MultiScreen® Filter Plates. Merck KGaA, Darmstadt, Germany
- Brochure (2022). MultiScreen® and Millicell® Plates for Assay Development. State-of-the-art tools for drug discovery research and sample preparation. Merck KGaA, Darmstadt, Germany
- Product flyer (2025). MultiScreen®HTS Glass Fiber Filter Plates. 96-well and 384-well plates incorporate optimized glass fiber for assays including receptor-ligand binding. Merck KGaA, Darmstadt, Germany
- Data sheet (2025). MultiScreen®HTS Filter Plates. Advanced plate design for full automation compatibility applications.Merck KGaA, Darmstadt, Germany
- Data sheet (2025). MultiScreen®HTS Filter Plates for ELISpot. Optimized filter plates measure immune response on a single cell level. Merck KGaA, Darmstadt, Germany
- Data sheet (2026). MultiScreen® MIC Plates. 96-well system for cell-based functional screening assays including migration, invasion and chemotaxis. Merck KGaA, Darmstadt, Germany
- Product page (Accessed March 16 2026). MultiScreen® Filter Plates. Sterile Lab Media Filtration & Cultureware. Merck KGaA, Darmstadt, Germany
- Chen C and Park J (2021) Application note. A highly sensitive IFN-γ ELISpot assay to quantify cellular immune responses to previous viral infection. Merck KGaA, Darmstadt, Germany
- Application note (2023) Development of a fluorescent ELISpot duplexing assay to measure immune response in cryopreserved PBMCs. Merck KGaA, Darmstadt, Germany