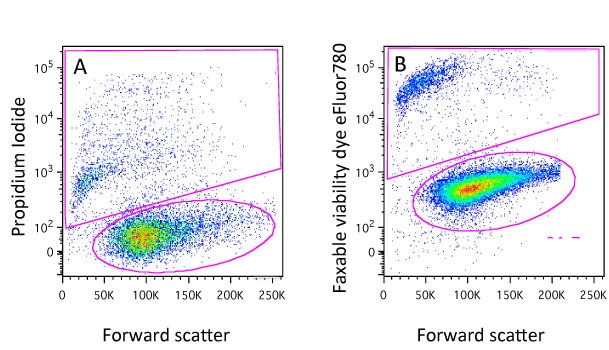
Flow cytometry is a fluorescence-based technology, as is fluorescence microscopy and confocal microscopy. Fluorescence is fundamental to how a cytometer gathers data, but I am often surprised, as a core manager, at how little new users know about the process of fluorescence. So, this is where I always start the training process.
Let’s get physical – how fluorescence works
The physical process of fluorescence occurs when a photon of light of a given wavelength hits a molecule and an electron is excited from a lower to higher energy level – this is unstable and the electron is losing energy via rotation and vibration. At some point, it will collapse back to ground state and release energy. Mostly the energy will be released as light – as a photon. At the point of emission, as some energy has been absorbed during the excitation process, the emitted light will also be of lower energy, i.e a longer wavelength. In this way, with fluorescence, we are always looking from the ultraviolet towards the infra-red range of the electromagnetic spectrum. This process of excitation and emission can be visualized in a Jablonski diagram. The process of excitation and emission takes only a few nanoseconds for most fluorochromes, but it is a stochastic process. Knowing this will help us interpret and understand the spectral diagram in Figure 1.
Figure 1. Excitation and emission spectra of FITC. Image created using ThermoFisher’s Spectral viewer.
Spectral viewers
All fluorochromes have an excitation spectrum (the dotted green line) – a range of wavelengths over which they can be excited – and an emission spectrum – (the solid trace) a range over which they emit. This all looks very simple but contains information that we need to be aware of. We need to know “does my cytometer have the appropriate excitation wavelength and collections filters for this fluorochrome”. Most manufacturers of cytometers and fluorescent dyes and reagents will have an online spectral viewer, and it’s worth bookmarking as many as possible because some dyes are proprietary and only appear on one website.
Exciting the dye!
We know that we can excite this fluorochrome (FITC or fluorescein isothicyanate) anywhere between 300 and 550nm, but we know, as this is a normalized curve, that we get maximum excitation around 595nm. So, we know if we have a light source in our cytometer close to this wavelength that we will get good excitation of this molecule. We also know that a common excitation wavelength found in cytometers is 488nm, so we should be able to excite this dye. But any wavelength of light within the excitation spectrum will excite the dye, just not as efficiently, so even our 405nm violet laser will excite FITC to a small degree. Being aware of this will help us with our flow experimental design and the interpretation of the data.
Where do the emitted photons fall?
The second part of the graph is the emission spectrum – this is the range of photons that will be produced by the fluorochromes. You may hear FITC referred to as being ‘green’ – we shouldn’t refer to things by color (but unfortunately even seasoned cytometrists still do!) but should rather refer to wavelength of the light, which is a more specific descriptor. Remember that the time scale of the process of moving an electron to a higher orbit and returning to ground state is uncertain, but what is certain is that while the electron is up there it is absorbing some of the energy of the exciting photon. So, when it does collapse back to ground state, it will release a photon that is of longer wavelength. Be aware that the energy is not always released as light. There are other processes by which the energy may be dissipated, e.g heat. Because of this uncertainty we don’t know the exact wavelength that will be emitted. But remember that this is a normalized curve. Most of the time FITC will emit photons around 530nm. Occasionally the emission process will be very rapid and a photon around 500nm will be released, and sometimes the electron will stay longer in the excited state, absorb more energy, and release a photon around 580nm (‘orange’) or even 650nm (‘red’).
This curve though allows us to know if the filters that we have in our cytometer would be able to detect light emitted. Again we know that a common bandpass filter used is 530/30 (meaning that light between 515nm and 545nm will pass through it. It also allows us to see whether photons from this dye would also be picked up in optical channels that are normally used for other dyes and would allow us to see if we would need to perform fluorescence compensation, which is needed in multicolor experiments
A list of the common spectral viewers: