Let me introduce you to ELISA…No, not the girl…The assay.
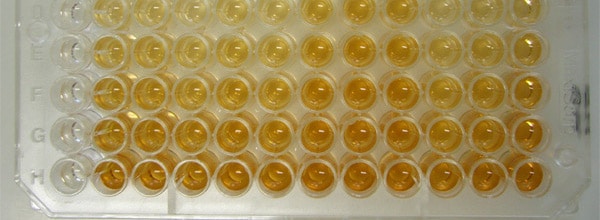
An ELISA (Enzyme-Linked ImmunoSorbant Assay) is a popular assay that uses antibodies and color change to detect proteins, peptides, antibodies or biomolecules in complex mixtures. ELISAs are popular because they are reliable, specific, easy to use, and can easily be scaled up to process multiple samples simultaneously.
How an ELISA is Done:
In an ELISA, your protein is first captured; and second, detected. There are two main ways (direct and indirect) that this protein capture and detection are done, see below.
Capture Way 1: Direct Capture ELISA
The way a direct capture ELISA works is similar to how proteins are captured in Western blotting. But in a direct capture ELISA, instead of immobilizing your protein on nylon or nitrocellulose membrane as you would in Western blotting, your protein is immobilized within a well of a microtiter plate. This is done by applying to your sample a “coating buffer” made of a carbonate-bicarbonate buffer (0.2 M sodium carbonate/bicarbonate pH 9.4) to directly immobilize your protein. It is important to note that this kind of capture is nonspecific. In fact when using a coating buffer the entire protein content of your complex mixture will be captured and coated onto the well of your plate.
Advantages/Disadvantages to Direct Capture:
This method is good if there is no commercial ELISA available for your protein-of-interest. But, because all proteins in your mixture are captured alongside your protein-of-interest, in direct capture ELISA you may experience higher background noise than if you used an indirect capture ELISA (see below).
Capture Way 2: Indirect Capture ELISA
In indirect capture ELISA, instead of capturing your entire complex mixture, only your protein-of-interest is selectively captured and immobilized to the well of your plate. This selective capture is accomplished by using an immobilized antibody at the bottom of your plate well to selectively bind and capture your protein-of-interest. An indirect capture ELISA is commonly referred to as a “sandwich ELISA” because when complete, your protein-of-interest will be sandwiched between two different antibodies – one antibody that is immobilizing it, and a second antibody that is detecting it (see below about detecting).
Advantages/Disadvantages to Indirect Capture:
This method is usually preferred, as it is more specific than the direct capture method. There are many commercial indirect Capture ELISA kits on the market, so start Googling.
Detection Way 1: Direct Detection ELISA
In direct detection a primary antibody conjugated to a reporter enzyme is bound to your immobilized protein. After excess (non-binding) antibody is rinsed away this reporter enzyme is then reacted with its substrate to produce a signal. This signal (often a color change or chemiluminescent signal) can then be detected and quantified using a spectrometer.
Advantages to Direct Detection:
- Quick. Only one antibody means fewer incubations and washes, making direct detection faster than indirect detection.
- Less cross reactivity. Since no secondary antibody is used, there is no chance of secondary antibody cross-reactivity.
Disadvantages to Direct Detection:
- Impaired immunoreactivity. Sometimes the immunoreactivity of the secondary antibody is impaired by its conjugation to the reporter enzyme. This can lower the sensitivity of a direct detection ELISA.
- Less flexible. You need an uniquely conjugated primary antibody for each protein-of-interest.
- No signal amplification. Unlike with indirect detection ELISAs, direct detection ELISAs have no signal amplification and are thus not as sensitive.
Detection Way 2: Indirect Detection ELISA
In an indirect detection ELISA, unlabeled primary antibodies are used to recognize your captured protein. This primary antibody is then, in turn, recognized by a conjugated secondary antibody. It is this secondary antibody conjugated to a reporter enzyme that is then reacted to a substrate to produce a colormetric signal, which is quantifiably by spectrometer.
Advantages to Indirect Detection:
- Sensitive. In an indirect detection ELISA, multiple conjugated secondary antibodies can bind to the primary antibodies. This means that multiple reporter enzymes can be bound to any one captured protein-of-interest. Thus an indirect detection method has signal amplification and is more sensitive than a direct detection method.
- Versatile. The same labeled secondary antibody can be used in numerous reactions.
- No impaired immunoreactivity. Because the primary antibody is not conjugated, it retains its full immonoreactivity, making indirect detection, again, more sensitive.
- Flexible. In an indirect detection ELISA, a variety of secondary antibody conjugates can be tried with any given primary antibody/protein combination. Thus it is easy to experiment with different visualization markers for any given primary antibody/protein combination.
Disadvantages to Indirect Detection:
- Cross-reactivity. Whenever you use a secondary antibody there is also the risk of cross-reactivity and a higher background signal.
- Slower. The use of two different antibodies means more incubations and washes. This makes an indirect detection ELISA slower.
Where an ELISA is Done
Most ELISAs are performed in flat bottom 96-well plates made of polysterene or polyvinyl. It is important when selecting a plate for an ELISA that you select a plate specially designed for an ELISA or other optical assays. These optical plates have been specially designed to have consistent optics, which will help you achieve more accurate and consistent results. Keep in mind also that if your ELISA produces a fluorescent or chemiluminescent signal, you should use an opaque (either black or white) microtiter plate.
Tips for Doing the Perfect ELISA
- In initial ELISA development it is important to check a range of parameters to establish the range of your assay.
- Temperature, buffers, humidity need to be kept consistent between samples and experiments.
- Make sure all pipettes are well calibrated and used properly.
- Consider using multichannel pipettes and automatic plate washers to minimize variability.
- Pay special attention to the wells at the edge of the plates. These wells are more vulnerable to error – due to evaporation, etc. To overcome this “edge effect” always test all samples in duplicate or triplicate.
- For more tips on performing a great ELISA see the articles: “Tips For Better ELISA Results” and “5 Ways to Improve Your ELISAs”.
- If things go wrong and you’re not sure why, you can get things on track by troubleshooting your ELISA.
Good Luck and Happy Protein Detecting!