Counting On Your Results – Tips And Technologies For Colony/Plaque Counting
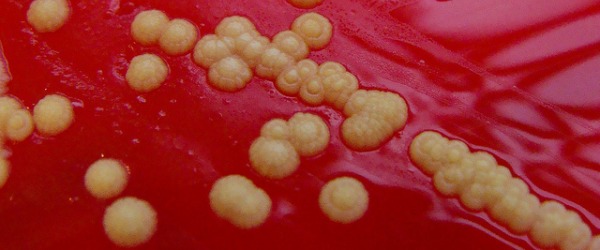
Have you ever emerged from the lab, bleary-eyed, blinking dazedly at the sun after spending hours hunched over a lab bench counting endless bacterial colonies or viral plaques?
A necessary evil…
I consider colony/plaque counting one of the necessary evils of working with microorganisms. Necessary because many experiments have an endpoint that requires determining the number of bugs present in a sample; evil because it can be tedious work that strains the eyes.
…but a simple one
A good thing about colony counting is that it is probably one of the simplest assays you will ever perform. It can also be one of the cheapest when performed manually. However, sometimes you are faced with mountains of plates to count and throwing some money and technology at the assay can significantly increase your speed (and save your poor eyes!).
Whichever way you approach colony/plaque counting, we are here to help. Below is a list of tips and a summary of available technologies for making sure you get accurate results efficiently.
Nice plates and accurate dilutions
It may seem silly, but starting with nice plates can make all the difference in an accurate count, whether being done by human or machine. Make sure your agar plates are smooth and free of bubbles. For plaques, make sure you have a nice even monolayer of cells with no clumps.
Even with the aid of technology it’s impossible to count hundreds of colonies/plaques in one dish. Likewise, having only one colony/plaque to count is meaningless. If you can’t predict the results of your experiment, plate several different dilutions- it will save you time in the end.
Tips for Manual Counting
If you are counting manually, make life a littler easier for yourself:
- Sit in a comfortable seat with lighting that illuminates the colonies/plaques the best. Putting the plates on a light box can be very helpful.
- Divide the plate into grids for ease of counting.
- Use a sharp-tipped marker to mark each colony/plaque as you count it.
- Use a handheld clicker-counter to help you keep track.
- To increase accuracy, count the plate, rotate 90 degrees and count again; or add a second person to confirm each plate’s tally.
- For a small investment (<$25; Cole-Parmer) you can buy a digital counter pen that records the number of marks you make according to pressure.
DIY Methods
If you are mechanically-inclined, you can put together your own automatic counter, as described by Brugger et al. Essentially you only need something to hold the plates, a camera, a light source and a computer. Other labs have used flatbed scanners to image plates. Once an appropriate image of the plate is captured, free software, (such as MATLAB which is available at www.coccus.eu) can be used to identify and count circular spots.
Commercial Counters
If you are faced with hundreds of plates to count each day, an automatic counter can make your life a lot easier. Automatic counters can range in cost from $4200-15,000. Basic models just count spots, while fancier models with high-res color cameras can count spots, measure zones of inhibition and provide information on size and shape of the spot. One word of caution: automatic counters can be led astray by imperfect agar, debris, unusual colonies or plaques or even writing on the plate, so it is always wise to validate the system.
Promega’s Colony Counter App
As if your iPhone hasn’t already become an indispensable part of your personal life, it can now be used for colony counting thanks to the Colony Counter App developed by Promega and available at the iTunes store (https://itunes.apple.com/us/app/promega-colony-counter/id620431249?mt=8). This handy app costs only $3.99 and is simple to use. Take a picture of your plate and the app will automatically count the number of spots. You can then refine the count by marking missed colonies or masking out false positives. The app can also automatically average a count across a set of plates.
With these great tips and knowledge of available technologies, you will have no problem saying “Count me in!” the next time you’re asked to count hundreds of plates!
1 Comments
Leave a Comment
You must be logged in to post a comment.
ImageJ.